
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
8th Edition
ISBN: 8220102895805
Author: Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 4, Problem 4.27UKC
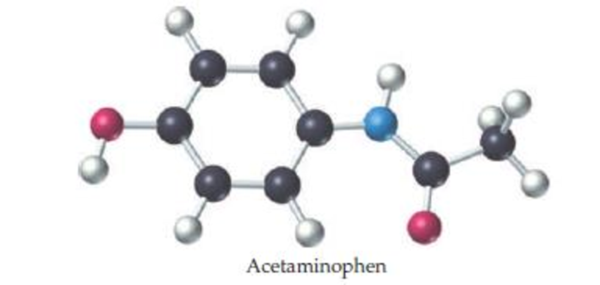
The ball-and-stick molecular model shown here is a representation of acetaminophen, the active ingredient in over-the-counter headache remedies such as Tylenol. The lines indicate only the connections between atoms not whether the bonds are single, double, or triple (red = O, gray = C, blue = N, ivory = H).
- (a) What is the molecular formula of acetaminophen?
- (b) Indicate the positions of the multiple bonds in acetaminophen.
- (c) What is the geometry around each carbon and each nitrogen?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Butadiene (right) is a colorless gas used to make synthetic rubber and many other compounds. (a) How many σ bonds and π bonds does the molecule have? (b) Are cis-trans arrangements about the double bonds possible? Explain.
Hypoglycin A, an amino acid derivative found in unripened lychee, is an acutely toxic compound that produces seizures, coma, and sometimes death in undernourished children when ingested on an empty stomach. (a) Draw the neutral, positively charged, and negatively charged forms of hypoglycin A. (b) Which form predominates at pH = 1, 6, and 11? (c) What is the structure of hypoclycin A at its isoelectric point?
Draw a structure for the compound, C3H5Br, that fits the following 1H NMR data:
δ 2.32 (3H, singlet)
δ 5.35 (1H, broad singlet)
δ 5.54 (1H, broad singlet)
Chapter 4 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
Ch. 4.1 - Prob. 4.1PCh. 4.2 - Prob. 4.2PCh. 4.2 - Prob. 4.3PCh. 4.3 - Prob. 4.4PCh. 4.3 - Prob. 4.5PCh. 4.4 - The BF3 molecule can also react with NH3 by...Ch. 4.5 - Prob. 4.7PCh. 4.7 - Prob. 4.8PCh. 4.7 - Add lone pairs where appropriate to the following...Ch. 4.7 - Prob. 4.10P
Ch. 4.7 - Prob. 4.11PCh. 4.7 - The molecular model shown here is a representation...Ch. 4.7 - Prob. 4.1CIAPCh. 4.7 - Prob. 4.2CIAPCh. 4.7 - Prob. 4.13PCh. 4.8 - Prob. 4.3CIAPCh. 4.8 - Prob. 4.4CIAPCh. 4.8 - Prob. 4.14PCh. 4.8 - Prob. 4.15PCh. 4.8 - Prob. 4.16PCh. 4.8 - Prob. 4.17KCPCh. 4.9 - The elements H, N, O, P, and S are commonly bonded...Ch. 4.9 - Prob. 4.19PCh. 4.10 - Look at the molecular shape of formaldehyde (CH2O)...Ch. 4.10 - Prob. 4.21PCh. 4.10 - Prob. 4.22KCPCh. 4.11 - Prob. 4.5CIAPCh. 4.11 - Prob. 4.6CIAPCh. 4.11 - Prob. 4.23PCh. 4.11 - Prob. 4.24PCh. 4 - What is the geometry around the central atom in...Ch. 4 - Prob. 4.26UKCCh. 4 - The ball-and-stick molecular model shown here is a...Ch. 4 - Prob. 4.28UKCCh. 4 - Prob. 4.29UKCCh. 4 - Prob. 4.30UKCCh. 4 - What is a covalent bond, and how does it differ...Ch. 4 - Prob. 4.32APCh. 4 - When are multiple bonds formed between atoms and...Ch. 4 - Identify the bonds formed between the following...Ch. 4 - Prob. 4.35APCh. 4 - Prob. 4.36APCh. 4 - Prob. 4.37APCh. 4 - Prob. 4.38APCh. 4 - Prob. 4.39APCh. 4 - Prob. 4.40APCh. 4 - Prob. 4.41APCh. 4 - Prob. 4.42APCh. 4 - Prob. 4.43APCh. 4 - Prob. 4.44APCh. 4 - Prob. 4.45APCh. 4 - Prob. 4.46APCh. 4 - Prob. 4.47APCh. 4 - If a research paper appeared reporting the...Ch. 4 - Consider the following possible structural...Ch. 4 - Prob. 4.50APCh. 4 - Prob. 4.51APCh. 4 - Prob. 4.52APCh. 4 - Prob. 4.53APCh. 4 - Prob. 4.54APCh. 4 - Draw a Lewis structure for the following...Ch. 4 - Prob. 4.56APCh. 4 - Ethanol, or grain alcohol, has the formula C2H6O...Ch. 4 - Prob. 4.58APCh. 4 - Tetrachloroethylene, C2Cl4, is used commercially...Ch. 4 - Prob. 4.60APCh. 4 - The carbonate ion, CO32, contains a double bond....Ch. 4 - Prob. 4.62APCh. 4 - Prob. 4.63APCh. 4 - Prob. 4.64APCh. 4 - Prob. 4.66APCh. 4 - Predict the geometry around each carbon atom in...Ch. 4 - Prob. 4.68APCh. 4 - Prob. 4.69APCh. 4 - Prob. 4.70APCh. 4 - Prob. 4.71APCh. 4 - Prob. 4.72APCh. 4 - Which of the following bonds are polar? If a bond...Ch. 4 - Prob. 4.74APCh. 4 - Based on electronegativity differences, would you...Ch. 4 - Arrange the following molecules in order of the...Ch. 4 - Prob. 4.77APCh. 4 - Prob. 4.78APCh. 4 - Prob. 4.79APCh. 4 - Prob. 4.80APCh. 4 - Prob. 4.81APCh. 4 - Prob. 4.82APCh. 4 - Prob. 4.83APCh. 4 - Prob. 4.84APCh. 4 - Prob. 4.85CPCh. 4 - Prob. 4.86CPCh. 4 - Prob. 4.87CPCh. 4 - Prob. 4.88CPCh. 4 - Prob. 4.89CPCh. 4 - The phosphonium ion, PH4+, is formed by reaction...Ch. 4 - Prob. 4.91CPCh. 4 - Prob. 4.92CPCh. 4 - Prob. 4.93CPCh. 4 - Prob. 4.94CPCh. 4 - Prob. 4.95CPCh. 4 - Prob. 4.96CPCh. 4 - Prob. 4.97CPCh. 4 - Write Lewis structures for molecules with the...Ch. 4 - Prob. 4.99CPCh. 4 - Prob. 4.100GPCh. 4 - Hydrazine is a substance used to make rocket fuel....Ch. 4 - Prob. 4.102GPCh. 4 - Titanium forms both molecular and ionic compounds...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify the acid on the left and its conjugate base on the right in the following equations:(a) HOCl + H2O ↔ H3O+ + OCl-(b) HONH2 + H2O ↔ HONH3+ + OH-(c) NH4+ + H2O ↔ NH3 + H3O+(d) 2HCO3-2 ↔ H2CO3 + CO3-2 (e) PO4-3 + H2PO4- ↔ 2HPO4-2arrow_forward(A) What are Waxes? Draw the structure of wax, which is made up of palmitic acid (16:0) and a saturated 18-carbon alcohol. (В) Consider the structure of menthol. How many isoprene units are present in menthol? (C) What type of isoprene linkage (head-to-tail or tail-to-tail) is present in menthol? Identify the isoprene linkage in the following structure of menthol and indicate by a circle. Menthol =arrow_forwardThe empirical formula of the sugar glucose is C6H12O6. (a) How many moles are there in 270 g of glucose? (b) Calculate the molarity of a solution of 324 g of glucose dissolved in 2.0 l of water.arrow_forward
- Draw structural formulas for all of the following. Q.) Alcohols with the molecular formula C4H10Oarrow_forwardThe much-abused drug cocaine is an alkaloid. Alkaloids are noted for their bitter taste, an indication of their basic properties. Cocaine, C17H21O4N, is soluble in water to the extent of 0.17g/100mL solution, and a saturated solution has a pH = 10.08. What is the value of Kb for cocaine?arrow_forwardDraw the complete structural formula of arachidonic acid (Table 23.1) in a way that shows the cis stereochemistry of its four double bonds.arrow_forward
- Give structures for the following, based on the data in Table . (a) cis-9-Dodecenoic acid (b) 18:1c∆11 (c) A saturated fatty acid that should melt below 30 °Carrow_forwardConsider nitrous acid, HNO2 (HONO).(a) Write a Lewis structure.(b) What are the electron pair and molecular geometries of the internal oxygen and nitrogen atoms in the HNO2 molecule?(c) What is the hybridization on the internal oxygen and nitrogen atoms in HNO2?arrow_forwardSpermaceti, a fragrant substance isolated from sperm whales, was commonly used in cosmetics until it was banned in 1976 to protect the whales from extinction. Chemically, spermaceti is cetyl palmitate, the ester of palmiticacid with cetyl alcohol (the straight-chain 16-carbon alcohol). Draw the structure of spermaceti.arrow_forward
- (b) The structure of a drug called tirbanibulin which is used to treat a pre-cancerous skin condition called actinic keratosis is shown below. (i) (ii) (iii) (iv) ہے مههممنن State Lipinski's Rule of Five (ROF) and Veber's addition to the ROF. Briefly state why the ROF is used in medicinal chemistry?. Given that the molecular mass of tirbanibulin is 431.536 g.mol-¹ and its log P is 3.2, apply the ROF and Veber's additional rule to this drug and show clearly how you arrived at your answer. Comment on the outcome of your analysis of the structure in part (iii).arrow_forwardDraw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic acidarrow_forwardConsider these compounds: A. PbBr, B. MnS C. Ag,CO3 D. AIPO, Complete the following statements by entering the letter(s) corresponding to the correct compound(s). (If more than one compound fits the description, include all the relevant compounds by writing your answer as a string of characters without punctuation, e.g, ABC.) Without doing any calculations it is possible to determine that magnesium fluoride is more soluble than and magnesium fluoride is less soluble than| It is not possible to determine whether magnesium fluoride is more or less soluble than by simply comparing Kgp values.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license