
Interpretation:
The value of
Concept introduction:
The Gibb’s free energy of mixing determines whether the process of mixing is spontaneous or not at a constant pressure and temperature. The expression to calculate the Gibb’s free energy of mixing is shown below.
Answer to Problem 4.76E
The graph between
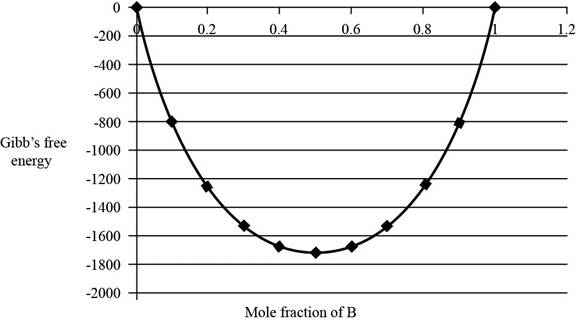
The value of
Explanation of Solution
The formula to calculate Gibb’s free energy of mixing is shown below.-
Where,
•
•
•
•
•
Conversion of temperature from Celsius to Kelvin is shown below.
Substitute the temperature
The temperature is
The mole fraction of gas A and gas B is equal to one as shown below.
Substitute the value of
The value of
Substitute the values of
The value of
Similarly, the value of
Therefore, the value of
The graph between
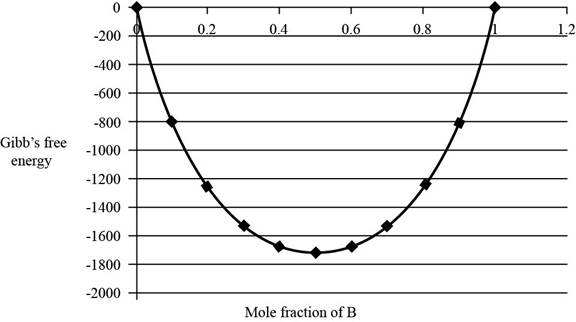
Figure 1
The graph between
Want to see more full solutions like this?
Chapter 4 Solutions
Physical Chemistry
- What is the sign of the standard Gibbs free-energy change at low temperatures and at high temperatures for the explosive decomposition of TNT? Use your knowledge of TNT and the chemical equation, particularly the phases, to answer this question. (Thermodynamic data for TNT are not in Appendix G.) 2C7H5N3O6(s) 3N2(g) + 5H2O() + 7C(s) + 7CO(g)arrow_forwardDetermine the standard Gibbs free energy change, rG, for the reactions of liquid methanol, of CO(g), and ofethyne, C2H2(g), with oxygen gas to form gaseous carbondioxide and (if hydrogen is present) liquid water at298 K. Use your calculations to decide which of thesesubstances are kinetically stable and which are thermodynamically stable: CH3OH(), CO(g), C2H9(g), CO2(g),H2O().arrow_forwardWhat is the sign of the standard Gibbs free-energy change at low temperatures and at high temperatures for the synthesis of ammonia? 3H2(g) + N2(g) 2NH3(g)arrow_forward
- Use data from Appendix D to calculate the standardentropy change at 25°C for the reaction CH3COOH(g)+NH3(g)CH3NH2(g)+CO2(g)+H2(g) Suppose that 1.00 mol each of solid acetamide, CH3CONH2(s), and water, H2O(l), react to give thesame products. Will the standard entropy change belarger or smaller than that calculated for the reactionin part (a)?arrow_forwardFor the reaction BaCO3(s) BaO(s) + CO2(g), rG = +219.7 kJ/mol-rxn. Using this value and other data available in Appendix L, calculate the value of fG for BaCO3(s).arrow_forwardAppendix J lists standard molar entropies S, not standard entropies of formation rS. Why is this possible forentropy but not for internal energy, enthalpy, or Gibbsfree energy?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





