
Concept explainers
(a)
Interpretation:
The following ball- and -stick model of 3-methyl-2-hexenoic acid should be converted into a Lewis structure.
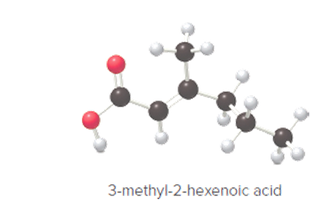
Concept introduction:
- Lewis structures are the diagrams that show the bonding between the atoms of the molecules and existing lone pairs of electrons.
- Bonding electrons are those electrons which are shared between the atoms resulting in the formation of bond.
- Non-bonding electrons are the valence electrons of the atom which are not shared with another atom.
(b)
Interpretation:
The shape around each carbon atom should be determined.
Concept introduction:
The model which is used in chemistry to determine the geometry of individual molecules with the help of the number of electron pairs surrounded by central atoms is known as Valence shell electron pair repulsion (VSEPR) theory.
(c)
Interpretation:
The polar bonds present in 3-methyl-2-hexenoic acid should be labeled.
Concept introduction:
A polar bond is a covalent bond between two atoms where the electrons forming the bond are unequally distributed. This causes the molecule to have a slight electrical dipole moment where one end is slightly positive and the other is slightly negative.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Using the symbols and +, indicate the direction of polarity in each polar covalent bond. (a) CN (b) NO (c) CClarrow_forwardAnswer the following. Covalent Compound Lewis Structure with correct geometry Geometry Name Polar or Nonpolar CH4 NH3 SF4 XeOF4 SF6arrow_forwardWrite a Lewis structure for each of the following molecules. H3COCH3 (oxygen is between the two carbon atoms) Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons.arrow_forward
- Draw an acceptable Lewis structure for each compound, assuming the atoms are connected asarranged. Hydrogen cyanide (HCN) is a poison, formaldehyde (H2CO) is a preservative, and glycolic acid (HOCH2CO2H) is used to make dissolving sutures.arrow_forwardDraw the correct Lewis structure for the following molecules so4-2 OH-arrow_forwardA. Draw the Lewis Dot Structure of the molecule or ion OF2 B. Find the shape of the molecule or ion OF2 C. Draw the Lewis Dot Structure of the molecule or ion CH2F2 D. Find the shape of the molecule or ion CH2F2 E. Draw the Lewis Dot Structure of the molecule or ion PO3^-3 F. Find the shape of the molecule or ion PO3^-3arrow_forward
- Identify any polar covalent bonds by drawing dipole arrows in the appropriate locations.arrow_forwardComplete Table 3 (Remember that the values pertain to the Central atoms only. In this table, you will have more than one central atom so report more than one value, ie. 3 / 3) Table 3 Molecules with Multiple Central Atoms Molecule C2H4 H2O2. CH3OH. CH3NH2 Number of Bond Pairs / / / / Number of Lone Pairs / / / / Number of Electron Domains / / / / Molecular Geometry (Shape) / / / / Bond Angle / / / / Polar or Non-Polar Nonpolar Polar Polar Polararrow_forwardDetermine whether each molecule is polar or nonpolar by writing either polar or nonpolar in each box.sulfur tetrachloride: phosphorus pentaiodide: sulfur hexafluoride: nitrogen tribromide: carbon monoxide:arrow_forward
- please check the screenshot fiind Lewis Structure (show all electrons and bonds) e– geometry Approx. Bond angles Polar or non-polar (don't worry about ions) Total lone pairs in the entire molecule Number of nonbonding electron pairs on the central atomarrow_forwardDraw a Lewis Structure For CH4O. Polar or nonpolar molecule? Draw also a 3D structure (entire molecule) for CH4Oarrow_forwardDecide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative side. For example, if the molecule were HCl and you decided the hydrogen atom was closest to the negative side of the molecule, you'd enter "H" in the last column of the table.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

