
Concept explainers
Figure 40-22 shows three points at which a spin-up electron can be placed in a nonuniform magnetic field (there is a gradient along the z axis). (a) Rank the three points according to the energy U of the electron’s intrinsic magnetic dipole moment
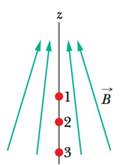
Figure 40-22 Question 8.
Want to see the full answer?
Check out a sample textbook solution
Chapter 40 Solutions
Fundamentals of Physics, Volume 1, Chapter 1-20
Additional Science Textbook Solutions
Physics for Scientists and Engineers with Modern Physics
Introduction to Electrodynamics
Glencoe Physics: Principles and Problems, Student Edition
Essential University Physics: Volume 2 (3rd Edition)
Conceptual Physics (12th Edition)
Lecture- Tutorials for Introductory Astronomy
- On what factors does the orbital magnetic dipole moment of an electron depend?arrow_forwardAt saturation, when nearly all the atoms have their magnetic moments aligned, the magnetic field is equal to the permeability constant 0 multiplied by the magnetic moment per unit volume. In a sample of iron, where the number density of atoms is approximately 8.50 1028 atoms/m3, the magnetic field can reach 2.00 T. If each electron contributes a magnetic moment of 9.27 10-24 A m2 (1 Bohr magneton), how many electrons per atom contribute to the saturated Held of iron?arrow_forwardThe force on a magnetic moment z in a nonuniform magnetic field Bz is given by Fz=zdBzdz If a beam of silver atoms travels a horizontal distance of 1 m through such a field and each atom has a speed of 100 m/s, how strong must the field gradient dBz/dz be in order to deflect the beam 1 mm?arrow_forward
- If an electron in an atom has an orbital angular momentum with ml = 2, what are the components (a) Lorb,z and (b) μorb,z? If the atom is in an external magnetic field that has magnitude 33 mT and is directed along the z axis, what are (c) the potential energy Uorb associated with the electron's orbital magnetic dipole moment and (d) the magnitude of the potential energy Uspin associated with the electron's spin magnetic dipole moment? If, instead, the electron has ml = -1, what are (e) Lorb,z, (f) μorb,z, (g) the potential energy Uorb associated with the electron's orbital magnetic dipole moment and (h) the magnitude of the potential energy Uspin associated with the electron's spin magnetic dipole moment?arrow_forwardThe magnetism of permanent magnets arises because the inherent magnetic moment of electrons causes them to act like little compass needles. Protons also have an inherent magnetic moment, and this is the basis for magnetic resonance imaging (MRI) in medicine.Although a compass needle would prefer to align with a magnetic field, the needle can point in any direction. This isn’t the case for the magnetic moment of a proton. Quantum physics tells us that the proton’s energy must be quantized. There are only two possible energy levels—and thus two possible orientations—for protons in a magnetic field: E1 = -μB magnetic moment aligned with the field E2 = +μB magnetic moment aligned opposite the fieldwhere μ = 1.41 x 10-26 J/T is the known value of the proton’s magnetic moment. FIGURE 28.24 shows the two possible energy states. The magnetic moment, like a compass needle, “wants” to align with the field, so that is the lower-energy state.…arrow_forwardIn a deuterium nucleus, the proton and neutron spins can be either parallel or antiparallel. What are the possible values of the total spin of the deuterium nucleus? (It is not necessary to consider any orbital angular momentum.) The magnetic dipole moment of the deuterium nucleus is measured to be nonzero. Which of the possible spins is eliminated by this measured value?arrow_forward
- Magnetization of a water sample. (a) Calculate the number of particles that contribute to the magnetization of a 2 cm3 sample of water at 37°C in a field strength of 3T. Remember the number of 1H nuclei in the water molecule. (b) Calculate the magnitude of the magnetization vector M for that sample.arrow_forwardThe potential energy of a magnetic moment in an external magnetic field is given by U = -u-B. The magnetic moment associated with the spin of an electron is 5.79 x 10-5 eV/T. Calculate the difference in energy between the two possible orientations of an electron in energy in a magnetic field B = (0.8 T) k. Answer in units of eV. %3D %3D If these electrons are bombarded with photons of energy equal to this energy difference, "spin flip" transitions can be induced. Find the wavelength of the photons needed for such transitions. (This phenomenon is called electron spin resonance.) Answer in units of cm.arrow_forwardF.j = (-e)(-v,k) × (B,i + B.k) ev Bzj. Solving for B gives F. Br 8.50x10 16 N = 1.13 T. Therefore B = 1.13 Ti – 0.772 Tk. The magnitude o A group of particles is traveling in a magnetic field of unknown magnitude and direction. You observe that a proton moving at 1.70 km/s in the +x-direction experiences a force of 2.10x10-16 N in the +y- direction, and an electron moving at 4.70 km/s in the eve (1.60x10 19 C)(4700 m/s) is B = VB? + B = (1.13 T)² + (-0.772 T)² = 1.37 T. -z-direction experiences a force of 8.50x10-16 N in the +y-direction. Part B For related problem-solving tips and strategies, you may want to view a Video Tutor Solution of Magnetic force on a proton. What is the direction of the magnetic field? (in the xz-plane) Express your answer in degrees. 0 = 60.59 ° from the -z-direction to the +x-direction Submit Previous Answers Request Answer X Incorrect; Try Again; 9 attempts remainingarrow_forward
- The Hall affect is used to measure the carrier density of a thin sheet of electrons. When a current of 80.0 µA flows through the length of the electron sheet, which is 1.2-mm long, 0.27- mm wide and 12-nm thick, a magnetic field perpendicular to the sheet produces a potential difference of 0.53 mV across the width of the sheet. If the carrier density in the electron sheet is 6.74 x 1025 m-3, what is the magnitude of the magnetic field?arrow_forwardIf the speed of the electron in Example 19-4 were 7.3 * 105m>s,what would be the corresponding orbital radius?arrow_forwardIf an electron in an atom has an orbital angular momentum with m = 4, what are the components (a) Lorb,z and (b) Horb,z? If the atom is in an external magnetic field that has magnitude 33 mT and is directed along the z axis, what are (c) the potential energy Uorb associated with the electron's orbital magnetic dipole moment and (d) the magnitude of the potential energy Uspin associated with the electron's spin magnetic dipole moment? If, instead, the electron has m₁ =-5, what are (e) Lorb,z, (f) Horb,z, (g) the potential energy Urb associated with the electron's orbital magnetic dipole moment and (h) the magnitude of the potential energy Uspin associated with the electron's spin magnetic dipole moment? (a) Number (b) Number (c) Number (d) Number (e) Number (f) Number (g) Number (h) Number Units Units Units Units Units Units Units Unitsarrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning



