
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.1, Problem 4.2P
Consider the following reaction, in which an
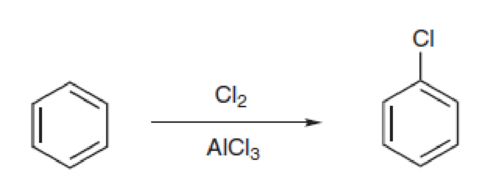
The mechanism is very similar to the mechanism for bromination. First, Cl2 reacts with AlCl3 to generate a complex that can serve as a source of Cl+. Draw a mechanism for formation of this complex.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
When diethyl ether (CH3CH2OCH2CH3) is treated with concentrated HBr, the initial products are CH3CH2Br and CH3CH2OH. Propose a mechanism to account for this reaction.
Draw and explain the mechanism in the reaction from compound 8, 9 to compound 10
e) NaH, RT, 72 h, 73%, >19:1 (E):(Z)
What is the mechanism for this series of reactions?
Chapter 4 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 4.1 - Consider the following reaction, in which an...Ch. 4.1 - Prob. 4.3PCh. 4.1 - Aromatic rings will also undergo iodination when...Ch. 4.2 - In each of the following cases, identify the...Ch. 4.2 - In each of the following cases, identify the...Ch. 4.2 - In each of the following cases, identify the...Ch. 4.3 - Prob. 4.10PCh. 4.3 - Prob. 4.11PCh. 4.3 - Prob. 4.12PCh. 4.3 - Prob. 4.13P
Ch. 4.3 - Prob. 4.14PCh. 4.3 - Predict the products of the following reaction.Ch. 4.3 - Prob. 4.16PCh. 4.3 - Prob. 4.17PCh. 4.4 - Identify the reagents you would use to achieve...Ch. 4.4 - Identify the reagents you would use to achieve...Ch. 4.4 - Identify the reagents you would use to achieve...Ch. 4.4 - Identify the reagents you would use to achieve...Ch. 4.4 - fill in the reagents you would use for the...Ch. 4.4 - fill in the reagents you would use for the...Ch. 4.4 - fill in the reagents you would use for the...Ch. 4.4 - fill in the reagents you would use for the...Ch. 4.4 - Prob. 4.27PCh. 4.4 - Prob. 4.28PCh. 4.4 - And now, for a challenging problem, try to draw...Ch. 4.6 - Prob. 4.31PCh. 4.6 - Prob. 4.32PCh. 4.6 - Prob. 4.33PCh. 4.6 - Prob. 4.34PCh. 4.6 - Prob. 4.35PCh. 4.6 - Prob. 4.36PCh. 4.6 - Prob. 4.37PCh. 4.6 - Prob. 4.40PCh. 4.6 - Prob. 4.41PCh. 4.6 - Predict the products for each of the following...Ch. 4.6 - Predict the products for each of the following...Ch. 4.6 - Predict the products for each of the following...Ch. 4.6 - Predict the products for each of the following...Ch. 4.6 - Prob. 4.47PCh. 4.6 - Prob. 4.48PCh. 4.6 - Prob. 4.49PCh. 4.6 - Prob. 4.50PCh. 4.6 - Prob. 4.51PCh. 4.6 - Prob. 4.52PCh. 4.6 - Prob. 4.53PCh. 4.6 - Prob. 4.54PCh. 4.6 - Prob. 4.55PCh. 4.6 - Prob. 4.56PCh. 4.7 - Prob. 4.58PCh. 4.7 - Prob. 4.59PCh. 4.7 - Prob. 4.60PCh. 4.7 - Prob. 4.61PCh. 4.7 - Prob. 4.62PCh. 4.7 - Prob. 4.63PCh. 4.7 - Prob. 4.64PCh. 4.7 - Prob. 4.65PCh. 4.7 - Prob. 4.66PCh. 4.7 - Prob. 4.67PCh. 4.7 - Can you explain why the following group is a...Ch. 4.7 - Prob. 4.70PCh. 4.7 - Prob. 4.71PCh. 4.7 - Prob. 4.72PCh. 4.7 - Prob. 4.73PCh. 4.7 - Prob. 4.74PCh. 4.7 - Prob. 4.76PCh. 4.7 - Prob. 4.77PCh. 4.7 - Prob. 4.78PCh. 4.7 - Prob. 4.79PCh. 4.8 - Propose an efficient synthesis for each of the...Ch. 4.8 - Propose an efficient synthesis for each of the...Ch. 4.8 - Propose an efficient synthesis for each of the...Ch. 4.8 - Propose an efficient synthesis for each of the...Ch. 4.8 - Propose an efficient synthesis for each of the...Ch. 4.8 - Prob. 4.87PCh. 4.8 - Prob. 4.88PCh. 4.8 - Prob. 4.89PCh. 4.8 - Prob. 4.90PCh. 4.8 - Prob. 4.91PCh. 4.8 - Prob. 4.92PCh. 4.9 - Prob. 4.94PCh. 4.9 - Prob. 4.95PCh. 4.9 - Prob. 4.96PCh. 4.9 - Prob. 4.97PCh. 4.9 - Prob. 4.98PCh. 4.9 - Prob. 4.99PCh. 4.9 - Prob. 4.100PCh. 4.9 - Prob. 4.101PCh. 4.9 - Prob. 4.102P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
In one stage of an annealing process, 304 stainless steelsheet is taken from 300 K to 1250 K as it passes throu...
Fundamentals of Heat and Mass Transfer
6.1 State the number of electrons that be must be lost by atoms of each of the following to achieve a stable el...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
17.25 You are asked to prepare a pH = 3.00 buffer solution starting from 1.25 L of a 1.00 M solution of hydrofl...
Chemistry: The Central Science (14th Edition)
Q7. What is the mass of a 1.75 L sample of a liquid that has a density of 0.921 g/mL?
a) 1.61 × 103 g
b) 1....
Chemistry: A Molecular Approach (4th Edition)
10.71 Identify each of the following as an acid or a base: (10.1)
H2SO4
RbOH
Ca(OH)2
HI
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Explain the mechanism of the reaction shown below.arrow_forwardThe conversion of cis-1,2-diphenylethene to trans-1,2-diphenylethene is catalyzed when I2 is added and the reaction mixture is irradiated with UV light. Provide a detailed mechanism for this reaction.arrow_forwardThe bicyclo [3.1.0] hexane ring system, highlighted in compound 3, is found in several natural products, including sabinene, a compound partially responsible for the flavor of ground black pepper. One method for preparing this ring system involves the conversion of compound 1 to compound 2, as shown below. Draw the structure of compound 2 and provide a reasonable mechanism for its formation. What are the most likely steps in the mechanism to form product 2? a. protonate, protonate, nucleophilic attack, nucleophilic attack b. Nucleophilic attack, protonate, protonate, nucleophilic attack c. Nucleophilic attack, protonate, nucleophilic attack, protonate d. Protonate, nucleophilic attack, protonate, nucleophilic attackarrow_forward
- Identify the products obtained when compound 1 is hydrolyzed in the presence of an acid and provide a detailed reaction mechanism for this hydrolysis of compound 1.arrow_forwardTo solve this problem, you need to read the description of the Hammett s, r treatment given in Chapter 15, Problem 92. When the rate constants for the hydrolysis of several morpholine enamines of para-substituted propiophenones are determined at pH 4.7, the r value is positive; however, when the rates of hydrolysis are determined at pH 10.4, the r value is negative.a. What is the rate-determining step of the hydrolysis reaction when it is carried out in a basic solution?b. What is the rate-determining step of the hydrolysis reaction when it is carried out in an acidic solution?arrow_forwardGive the full name of the reactions and its mechanism for each step.arrow_forward
- Draw reaction mechanism for week 2 A wittig reaction experiment: Week 1 -》 preparation of phosphonium salt Materials : triphenylphosphine (5.3g),methyl Bromoacetate 3.36g (2.1ml) , ethanol 30ml . The yield obtained was 8.93g .The percentage yield was 106.4% Week 2 -》 Formation of the ylide and wittig reaction Materials :Phosphonium salt (5g), napthyl-2- carboxaldehyde( 2.65g), 20 ml of water and 5 nl of Nahco3. Yield obtained was 0.51g . The percentage yield was 14.2%. Week 3: Solvent free wittig reaction Materials: Benzyltriphenylphosphonium chloride (0.5g ), 4 - Bromoabenzaldehyde (0.24g), Potassium phosphate (tribasi c) 0.275g .The yield obtained was 1.21g. The percentage yield was 35.6%.arrow_forwardUsing Blocking Groups to Control the Regiochemical Outcome of an Electrophilic Aromatic Substitution Reaction (Identify, draw the mechanism and explain the reagents that will achieve the following transformation)arrow_forwardFor each reaction, give the expected substitution product, and predict whether the mechanism will be predominantly first order (SN1) or second-order (SN2). (a) 1@iodo@1@methylcyclohexane + ethanolarrow_forward
- Identify the mechanistic pathways, respectively, for the products in the following reaction. a) E1, SN1 b) E1, SN2 c) E2, SN1 d) E2, SN2arrow_forwardWhich mechanism is most efficient to synthesize this flavor on an industrial scale and why?arrow_forwarda. Propose a mechanism for the following reaction: b. Given that ΔH° for the reaction is -42 kcal/mol and the bond dissociation enthalpies for the C¬H, C¬Cl, and O¬H bonds are 101, 85, and 105 kcal/mol respectively, calculate the bond dissociation enthalpy of the O¬Cl bond.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License