
Interpretation:
Major product for the given reaction has to be predicted. The given reaction is shown as,
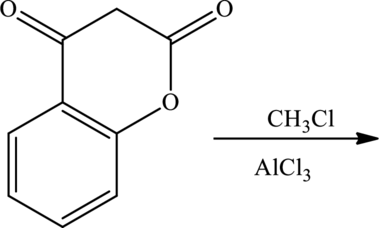
Concept Introduction:
Deactivators are electron withdrawing groups attached to the benzenes that have either positive charge or an atom with high electronegativity. They are meta directors.
Activators are electron donating groups attached to the benzenes that have either electron density that is able to push into benzene ring or a lone pair of electrons. They are ortho-para directing.
Halogens are deactivators that are ortho-para directing.
Rules to identify the group that dominates the directing effects:
- Ortho-para directors will always dominate meta directors.
- Strong activators will always dominate weak activators
Friedel-Crafts Alkylation: This Lewis acid-catalyzed electrophilic
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
- What are the products of each reactions below?arrow_forwardPredict the major products of the following reactions, including stereochemistry where appropriate. (a) sodium ethoxide + 1-bromobutane (b) sodium ethoxide + 2-methyl-2-bromobutanearrow_forwardPredict the product of each reaction below and indicate if the mechanism is likely to be SN1, SN2, E1, E2, or E1arrow_forward
- Predict the major product(s) for each of the following reactions. If no reaction is expected,write NR.arrow_forwardPredict the major product from the following reaction above. There might be more than one correct choice. Show the mechanism to prove your answer.arrow_forwardPredict the major products of the following reactions. p@methylanisole + acetyl chloride + AlCl3arrow_forward
- Predict the products of the following reactions: [PtCl4]2- + NO2- → (a) (a) + NH3 → (b) [PtCl3NH3]- + NO2- →(c) (c) + NO2- → (d) [PtCl(NH3)3]+ + NO2- → (e) (e) + NO2- → (f) [PtCl4]2- + I- → (g) (g) + I- → (h) [PtI4]- + Cl- →(i) (i) + Cl- → (j)arrow_forwardPredict the products of the following reactions. Include stereochemistry when necessary. For reactions with more than one step show the product formed after each step.arrow_forwardPredict the major products of the following reactions.(a) (R)-2-hexyl tosylate + NaCN(b) the tosylate of cyclohexylmethanol + excess NH3arrow_forward
- What is the expected product for the reaction below?arrow_forwardPredict the products of the following reaction. Include stereochemistry when necessary. For reactions with more than one step show the product formed after each step.arrow_forwardPropose mechanisms and show the expected products of the following reactions. p@nitrobromobenzene + methylamine (CH3 ¬NH2)arrow_forward
