
Concept explainers
(a)
An expression for the probability that an electron in ground state will be found outside a sphere of radius
(a)
Answer to Problem 78AP
The expression for the probability that an electron in ground state will be found outside a sphere of radius
Explanation of Solution
Write the expression for the radial probability density function for the Hydrogen atom in ground state.
Here,
Write the expression to find the probability to for an electron in grounds state to be found outside a sphere of radius
The probability outside a sphere is to be found. So integration must be done from radius of sphere to infinity.
Use expression (I) in (III) to find
Integrate expression (IV) by integration by parts.
Simplify expression (V) to find
Conclusion:
Apply limits from
Therefore, the expression for the probability that an electron in ground state will be found outside a sphere of radius
(b)
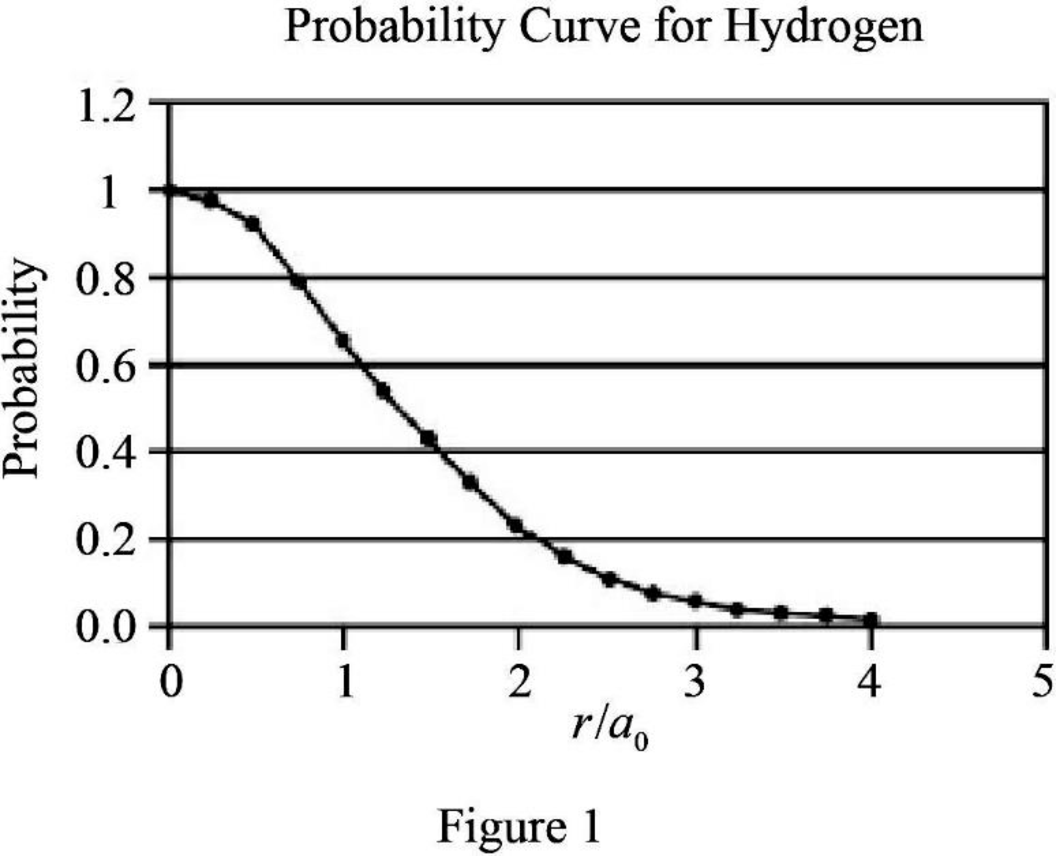
Graph the relation between probability and
(b)
Answer to Problem 78AP
The graph between probability and
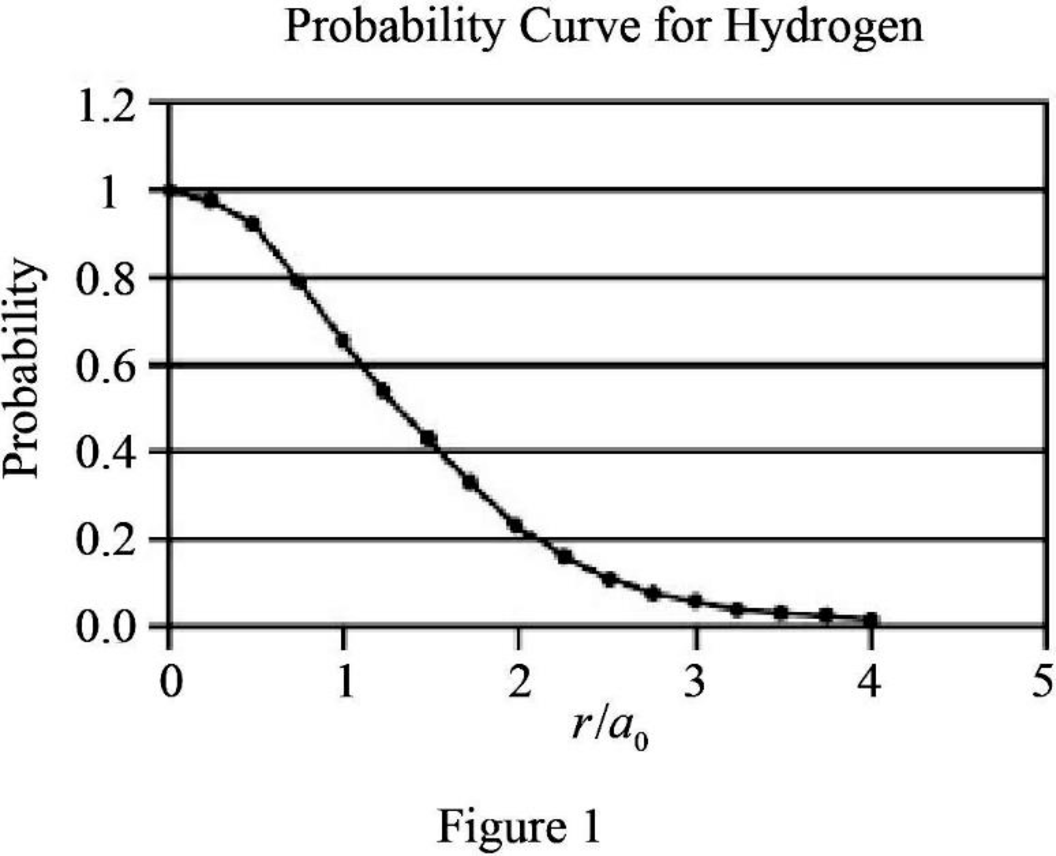
Explanation of Solution
The expression for probability as a function of
The expression is exponential in nature. The graph will be exponentially decreasing as the value of
Assume values between
Figure 1 below shows the plot between probability and
Conclusion:
Therefore, The graph between probability and
(c)
The value of
(c)
Answer to Problem 78AP
The value of
Explanation of Solution
The probability to detect an electron inside or outside a sphere of radius
Use expression for probability derived in part (a).
Substitute
Put
Conclusion:
Equation (VIII) is a transcendental equation. Solving it the value of
Therefore, the value of
Want to see more full solutions like this?
Chapter 42 Solutions
Bundle: Physics for Scientists and Engineers with Modern Physics, Loose-leaf Version, 9th + WebAssign Printed Access Card, Multi-Term
- The distance r between the electron and the nucleus in a hydrogenatom (in its lowest energy state) is a random variable with probability density p(r) = 4a03r2e- 2r/ao for r > 0, where ao is the Bohr radius (Figure 8). Calculate the probability P that the electron is within one Bohrradius of the nucleus. The value of a0 is approximately 5.29 x 10- 11 m,but this value is not needed to compute P.arrow_forwardChapter 38, Problem 071 For the arrangement of Figure (a) and Figure (b), electrons in the incident beam in region 1 have energy E has a height of U1 = 823 ev and the potential step = 617 ev. What is the angular wave number in (a) region 1 and (b) region 2? (c) What is the reflection coefficient? (d) If the incident beam sends 5.29 x 105 electrons against the potential step, approximately how many will be reflected? V= 0 V< 0 x = 0 region 1 region 2 (a) Energy --E- Electron (b)arrow_forwardConsider the function f(z) = 2 sin (E (z - 4) + 4 State the amplitude A, period P, and midline. State the phase shift and vertical translation. In the full period [0, P], state the maximum and minimum y-values and their corresponding z-values. Enter the exact answers. Amplitude: A = Number Period: P = sin (a) Midline: y = Number The phase shift is let e L The vertical translation is cha te tae Hints for the maximum and minimum values of f (z): The maximum value of y = sin (I) is y= 1 and the corresponding z values are z = 공 and multiples of 2 x less than and more than this z value. You may want to solve (z - 4) = • The minimum value of y = sin (z) is y = -1 and the corresponding z values are z = 폭 and multiples of 2 less than and more than this z value. You may want to solve (z - 4) = . • If you get a value for z that is less than 0, you could add multiples of P to get into the next cycles. • If you get a value for z that is more than P, you could subtract multiples of P to get into…arrow_forward
- If the radius of a calcium ion is 0.19 nm, how much energy does it take to singly ionize it? Give your answer in electron-volts (eV) with precision 0.1 eV. Give your answer to 2 significant digits. (with step pls)arrow_forwardA triply ionised beryllium atom (Be+++, Z = 4) has only one electron in orbit about the nucleus. If the electron decays from the n = 3 level to the first excited state (n = 2), calculate the wavelength of the photon emitted. give your answer in units of nm, rounded to one decimal place.arrow_forwardThe population density, Ni, corresponding to a discrete energy level, E₁, for a group of N like particles in Local Thermodynamic Equilibrium (LTE) state can be described by the following equation N₂ 9₁c-Ei/(KRT) Z(T) N i) Define the remaining quantities or constants in the above equation. ii) = Produce an expression for Z(T) as a function of T. In order to calculate Z(T) for a particular atomic gas such as argon, what atomic data or information needs to be made available before the calculation is carried out? iii) To uniquely describe the population density distribution corresponding to different discrete energy levels of a diatomic molecular gas such as CO in equilibrium, how many Z(T) functions need to be used and why?arrow_forward
- A triply ionised beryllium atom (Be+++, Z = 4) has only one electron in orbit about the nucleus. If the electron decays from the n 7 level to the first excited state (n = 2), calculate the wavelength of the photon emitted. Please give your answer in units of nm, rounded to one decimal place. Answer:arrow_forwardThe emergence of line spectra from a gas when a current passes through it was an observed phenomenon waiting for an explanation in the early 20th century. The atomic line spectra coming from elements such as hydrogen had been analyzed since the late 19th century. By studying the wavelength of the emerging radiation of hydrogen, experimenters found (often by trial and error) that the wavelengths in those spectra were described by the formula 1 = R ( 1/2 - 1/2 ) where R is known as the Rydberg constant. It has a value of R=1.097 × 107 m-¹. The variables n₁ and no are integer numbers (n₁ = 1, 2, 3, 4, ...). As experiments continued, scientists began to see more and more characteristic lines emerging from the hydrogen spectrum. Each of them corresponded to a wavelength predicted by this formula with some integer values for m₁ and ₂. In 1913, Niels Bohr provided an explanation for the observations made in experiments by proposing that each electron in an atom had only certain allowable…arrow_forwardAssume that the nucleus of an atom can be regarded as a three-dimensional box of width 2:10-¹4 m. If a proton moves as a particle in this box, find (a) the ground-state energy of proton in MeV and (b) the energies of the first excited state. (c) What are the degenerates of these states? Constants: h = 6.626-10-34 [J-s], m = 1.673-10-27 [kg] and ħ=h/2π.arrow_forward
- The ground-state wave function of a hydrogen atom is: 1 where r is distance from the nucleus and a, is the Bohr radius (53 pm). Following the Born approximation, calculate the probability, i.e., |w?dz, that the electron will be found somewhere within a small sphere of radius, ro, 1.2 pm centred on the nucleus.arrow_forwardFind the most portable value of r characterizing the distance between an electron in 2px and the nucleus in the He+ion. Discuss the physical meaning of the results.arrow_forwardConsider hydrogen in the ground state, 100 . (a) Use the derivative to determine the radial position for which the probability density, P(r), is a maximum. (b) Use the integral concept to determine the average radial position. (This is called the expectation value of the electrons radial position.) Express your answers into terms of the Bohr radius, a0. Hint: The expectation value is the just average value, (c) Why are these values different?arrow_forward
 University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning
Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning




