
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
6th Edition
ISBN: 9781305687875
Author: Gilbert
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 4.6, Problem 7E
Interpretation Introduction
Interpretation:Number of five-carbon units with below skeletal structure in geranial and vitamin A should be determined.
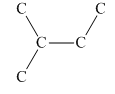
Concept introduction:Terpenes are varieties of organic compounds that are synthesized by plants. These are hydrocarbons that can be found in several plants and trees. These act as constituents for different oils.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For the Organic Chemistry topic of R and S, to make the structure R, does the prioritizing number have to be in order from 1 -> 2 -> 3 clockwise or can it be clockwise from 3 -> 2 -> 1?
Some natural proteins are rich in disulfide bonds, and their mechanical properties (tensile strength, viscosity, hardness, etc.) are correlated with the degree of disulfide bonding. (a) Glutenin, a wheat protein rich in disulfide bonds, is responsible for the cohesive and elastic character of dough made from wheat flour. Similarly, the hard, tough nature oftortoise shell is due to the extensive disulfide bonding in its α-keratin. What is the molecular basis for the correlation between disulfide-bond content and mechanical properties of the protein?(b) Most globular proteins are denatured and lose their activity when briefly heated to 65 °C. However, globular proteins that contain multiple disulfide bonds often must be heated longer at higher temperatures to denature them. One such protein is bovine pancreatic trypsin inhibitor (BPTI), which has 58 amino acid residues in a single chain andcontains three disulfide bonds. On cooling a solution of denatured BPTI, the activity of the…
Identify and justify the approximations used in the Huckel theory of conjugated hydrocarbons.
Chapter 4 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
Ch. 4.2 - Prob. 1ECh. 4.2 - Prob. 2ECh. 4.2 - Prob. 3ECh. 4.2 - Prob. 4ECh. 4.2 - Prob. 5ECh. 4.2 - Prob. 6ECh. 4.2 - Prob. 7ECh. 4.2 - Prob. 8ECh. 4.2 - Prob. 9ECh. 4.3 - Prob. 1E
Ch. 4.3 - Prob. 2ECh. 4.3 - Prob. 3ECh. 4.3 - Prob. 4ECh. 4.3 - Prob. 5ECh. 4.3 - Prob. 6ECh. 4.3 - Prob. 7ECh. 4.3 - Prob. 8ECh. 4.4 - Prob. 1ECh. 4.4 - Prob. 2ECh. 4.4 - Prob. 3ECh. 4.4 - Prob. 4ECh. 4.4 - Prob. 5ECh. 4.4 - Prob. 6ECh. 4.4 - Prob. 7ECh. 4.4 - Prob. 8ECh. 4.4 - Prob. 9ECh. 4.4 - Prob. 11ECh. 4.4 - Prob. 12ECh. 4.4 - Prob. 13ECh. 4.4 - Prob. 14ECh. 4.6 - Prob. 1ECh. 4.6 - Prob. 2ECh. 4.6 - Prob. 3ECh. 4.6 - Prob. 4ECh. 4.6 - Prob. 5ECh. 4.6 - Prob. 6ECh. 4.6 - Prob. 7ECh. 4.6 - Prob. 8ECh. 4.6 - Prob. 9E
Knowledge Booster
Similar questions
- Please help me with the organic chemistry question below. The solutions are linked, as it is one question with 2 parts: 1. A. Draw a molecular orbital diagram for (2E,4E)hexa-2,4-diene and ethene below. Make sure to label orbitals, HOMO, LUMO, nodes, antibonding/bonding, and fill correctly with electrons. 1. B. Use the orbitals drawn in the previous problem to determine if a cycloadditon would occur between the two compounds mentioned and if so, would it be suprafacial or antarafacial. Be sure to specify which orbitals you are using and use those orbitals to demonstrate your answer.arrow_forwardThe molecule shown on the right in the example in the right column is the amino acid histidine, and the five-membered ring is known as aromatic. An aromatic ring has 2, 6, 10, 14, etc., electrons placed in 2p orbitals around a ring. Indicate which of the following statements must therefore be true. 1. There are a total of six electrons in the pi system (defined as electrons in 2p orbitals), including the lone pair on the ring N that is not circled. 2. There are a total of six electrons in the pi system, including the lone pair on the ring N atom that is circled. 3. The lone pair on the ring N atom that is not circled resides in an sp2 orbital on an sp2 hybridized nitrogen atom. 4. Statements 2 and 3 are both truearrow_forwardI know that the answer to this question is 60, but I am struggling to understand why. Could somebody please draw this out for me 3-dimensionally or explain to me in detail why this is the case? One explanation I've heard is that it is because when the dihedral angle between the two methyl groups is 60, the molecular dipole moment between the two chlorine atoms is the smallest, therefore the answer is 60. This explanation is meaningless to me - that does not even seem to be what the question is asking. Is it a poorly-worded question? i need good explanationarrow_forward
- Measure the C-C-C and C-C-H bond angles in the energy-minimized models of cis and trans isomers of cyclooctene. Compare these values with those predicted by VSEPR. In which isomer are deviations from VSEPR predictions greater?arrow_forwardI ONLY NEED THE ANSWERS OF QUESTIONS 4, 5 and 6!! Explain briefly and clearly the following concepts, taking as reference the molecule of n-butane and the corresponding drawings or illustrations. See pages 149-152 of the book Organic Chemistry, sixth edition (J. G. Smith). 1. Potential energy of a conformation. 2. Dihedral angle (in chemical terms, nothing to do with planes) in relation to a rotatable bond for the molecule of interest. Present an example to illustrate the concept 3. What are the van der Waals interaction forces in a conformation? Then provide an illustration: 4. What is steric hindrance in a conformation? Then draw a picture to illustrate the concept? 5. What is the torsional stress of a conformation? Then draw a picture to illustrate the concept? 6. Describe 1,3-diaxial interaction and illustrate with a specific example.arrow_forwardThoroughly describe what is meant when a compound is considered aromatic. In your answer, provide examples and be sure to discuss how Huckel's Rule applies, and what it means to be anti-aromatic versus non-aromatic. When providing specific examples to demonstrate the requirements that you list, describe the delocalized pi electrons and sp2 hybridization. Lastly, why is benzene so special compared to alkenes like cyclohexene....both are 6-carbon atom cycles so are they chemically similar or not really similar at all?arrow_forward
- Which of the following compounds are consistent with the Huckel rule?arrow_forwardPredict the electronic configurations of (a) the benzeneanion, (b) the benzene cation. Estimate the π-bond energy in each case.arrow_forwardSteroids are another class of important biological molecules with a general substructure that contains three six membered rings fused to one another, ending in a fusion of a five-membered ring. Consider the structure shown below and use to : A.Convert the ring structure to a chair structure B. Identify each ring fusion as cis or trans and explain how you made that determinationarrow_forward
- Question:What is the significance of the LUMO (Lowest Unoccupied Molecular Orbital) in organic chemistry, and how does it contribute to chemical reactions?arrow_forwardGive the answer of this MCQ with detailed explanation in clear handwritten!arrow_forwardShow how to construct the molecular orbitals of ethylene, butadiene, and the allylicsystem. Show the electronic configurations of ethylene, butadiene, and the allylcation, radical, and anion.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning