
(a)
Interpretation:
Lewis structure of dimethyl ether has to be drawn.
Concept introduction:
Electron-dot (Lewis) symbol: The numbers of valence electron are shown using dot symbol around the atomic symbol.
The valance electrons are represented as a dot; the dots are distributed around the four sides of the
(b)
Interpretation:
The structure of diethyl ether has to be drawn and number carbon is calculated in diethyl ether.
(c)
Interpretation:
Interpretation:
A common structural feature of ether has to be explained.
Concept introduction:
Ethers:
An oxygen atom connected to two alkyl or aryl groups is called as ether. The general formula of ether is R–O–R′, where R and R′ group either alkyl or aryl groups.
Ethyl alcohol is reaction with acid which yields diethyl ether which is shown below.
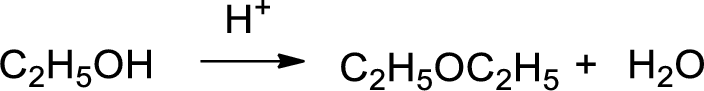
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Chemistry In Context
- Your roommate, a chemistry major, claims to have synthesized the compound CH5 in the lab. Why is that not possible?arrow_forwardWhat mass of water must be used to make 1.500 kg of 3.000 % ethanol?arrow_forwardConsider methane, CH₄, and hydrogen, H₂, as possible fuel sources a. Describe how methane and hydrogen could be obtained. Which of these methods do you think is less expensive? Explain. b. Which fuel do you think is more environmentally friendly? Explain.arrow_forward
- Which functional group or groups are not present in the compound shown in Figure 1. * A- Alcohol B- Amide C- Ether D- Alkyne E- All these functional groups are present in the compound.arrow_forwardConsider the molecule tetrahydrocannabinol (?21?30?2), or THC To be legal at the federal level, CBD oil may have no more than 0.3% THC by mass. (That is, a 1-g sample of CBD oil may contain no more than 3 mg of THC.) Assuming the density of CBD oil is 0.95 g/mL, what is the maximum number of moles of THC that may legally be present in 100-mL of CBD oil?arrow_forwardHow is the process of producing purified water?like a water station refill using deep well source of water?Step by step and what machine or equipment to used and what is the porpuse of that machine or equipment of producing purified water?arrow_forward
- What is natural gas? a. a gas that forms from decaying vegetation b. a mixture of hydrocarbon gases, mostly methane c. a gas that forms naturally from coal d. naturally occurring hydrogenarrow_forwardWhy are ducks waterproof? It’s because they produce copious amounts of oils from their uropygial glands and spread it across their feathers. In this exercise, we’ll be investigating the molecular structure of one of these preen oils to determine how it keeps ducks dry. Q.5 - Preen oil is actually a complicated mixture of many different organic compounds, such as the structure seen previously.. Ornithologists have determined that birds often use preen oil compounds for scent recognition. Below, several different chemicals isolated from preen oil are shown, along with their vapor pressures at room temperature. p-cymene has the highest vapor pressure, meaning it is the most easily evaporated compound of the three listed. Explain why p-cymene has a higher vapor pressure at room temperature compared to the other compounds. Make sure to explain what holds the p-cymene in the sample. (Image attached)arrow_forwardIs there any difference between 2-methylpropane and isobutane? Why is the structure in the image not named 2-methylpropane but isobutane. What is meant by "iso"?arrow_forward
- 4. What is the functional group found in this organic compound?arrow_forwardExplain the meaning of the word “organic” when used to describe certain foods. How is it related to the term “organic” when used with organic chemistry?arrow_forward(a) When the metallic element sodium combines with the nonmetallic element bromine, Br2(l), how can you determine the chemical formula of the product? How do you know whether the product is a solid, liquid, or gas at room temperature? Write the balanced chemical equation for the reaction. (b) When a hydrocarbon burns in air, what reactant besides the hydrocarbon is involved in the reaction? What products are formed? Write a balanced chemical equation for the combustion of benzene C6H6(l), in air.arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning




