
Batch processes are often used in chemical and pharmaceutical operations to achieve a desired chemical composition for the final product and typically involve a transient heating operation to take the product from room temperature to the desired process temperature. Consider a situation for which a chemical of density
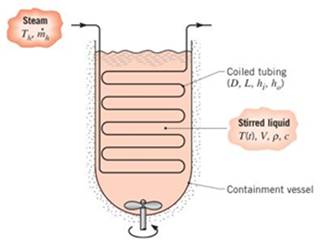
If the chemical is to be heated from 300 to 450 K in 60 min, what is the required length L of the submerged tubing?
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Fundamentals of Heat and Mass Transfer
Additional Engineering Textbook Solutions
Applied Statics and Strength of Materials (6th Edition)
Engineering Mechanics: Statics & Dynamics (14th Edition)
Automotive Technology: Principles, Diagnosis, and Service (5th Edition)
Degarmo's Materials And Processes In Manufacturing
Mechanics of Materials (10th Edition)
Introduction to Heat Transfer
- Two homogeneous spheres of mass M radius R joined by a bar of length L = 2R, negligible mass, and suspended from its geometric center by a rope such that when rotated ϕ exerts torque −kϕ with k torsion coefficient of the wire. The air exerts dissipative effect F = −6πηv on each sphere, where η is the viscosity of the air and v is the linear velocity of each sphere. If I0 = 25MR2 is the moment of inertia of a sphere with respect to its center of mass (image 1)determine the equation of motion of the pendulum. (image 2)arrow_forwardA 235-g sample of a substance is heated to 320 ∘C and then plunged into a 105-g aluminum calorimeter cup containing 195 g of water and a 17-g glass thermometer at 12.5 ∘C. The final temperature is 35.0∘C. The value of specific heat for aluminium is 900 J/kg⋅C∘ , for glass is 840 J/kg⋅C∘ , and for water is 4186 J/kg⋅C∘. What is the specific heat of the substance? (Assume no water boils away.)arrow_forwardA mass of 300kg of oil is cooled in 1 hour from 70 celcius to 35 celcius in a cooler consisting of a bank of tubes through which hot oil passes. Cooling water circulates around the outside of the tubes. Calculate the mass of cooling water required per hour if the water temp increases by 21 degrees celcius . Specific Heat of Oil - 2.0 kJ/kg deg C Specific Heat of Water - 4.186 kJ/kg deg Carrow_forward
- A long 8-cm diameter vertical steam pipe whose external surface temperature is 90 degrees C passes through some open area that is not protected against the wind blows. Determine the rate of heat loss from the pipe per unit length when the air is at 1 atm, 7 degrees Celsius and the wind is blowing across the pipe at a velocity of 50 km/h. From Table A-1:Thermal conductivity, k = 0.0275 W/m degrees Celsius ,viscosity, v = 1.77 x 10^-5 m^2/sPrandtl no. Pr = 0.71arrow_forwardA monatomic ideal gas initially fills a V0 = 0.45 m3 container at P0 = 85 kPa. The gas undergoes an isobaric expansion to V1 = 0.75 m3. Next it undergoes an isovolumetric cooling to its initial temperature T0. Finally it undergoes an isothermal compression to its initial pressure and volume. (a). Calculate the work done by the gas, W1, in kilojoules, during the isobaric expansion (first process). (b). Calculate the heat absorbed Q1, in kilojoules, during the isobaric expansion (first process). (c) Calculate the heat absorbed Q2, in kilojoules, during the isovolumetric cooling (second process). (d) Calculate the change in internal energy by the gas, ΔU2, in kilojoules, during the isovolumetric cooling (second process). (e) Calculate the work done by the gas, W3, in kilojoules, during the isothermal compression (third process). (f) Calculate the heat absorbed Q3, in kilojoules, during the isothermal compressions (third process).arrow_forwardthe title of the book: Engineering Thermofluids Thermodynamics, Fluid Mechanics, and Heat Transfer by: Mahmoud Massoudarrow_forward
- In the United States, energy for household heating is generally sold using English units, e.g., therm, gal, and cord. A house in Wisconsin uses 1200 therms of thermal energy during the heating season. Calculate the cost of fuel if the furnace uses (a) natural gas with an efficiency of 70%; (b) No. 2 fuel oil, efficiency 65%; (c) kerosene, efficiency 99.9% (unvented); and (d) wood with l5% moisture with an efficiency of 50%. Use the data in Tables 2.2, 2.7, and 2.13. The efficiencies are based on the HHV. Assume the cost of natural gas is $8/MBtu, the cost of No. 2 fuel oil is $3/gal, the cost of kerosene is $3.50/gal, and the cost of wood is $100/cord. Assume the bulk density of cord wood is 30 lbm/ft3 .arrow_forwardPressure cookers, in general, maintain a gage pressure of 2 atm (or 3 atm absolute) inside. Therefore, pressure cookers cook at a temperature of about 133°C instead of 100°C, cutting the cooking time by as much as 70 percent while minimizing the loss of nutrients. The newer pressure cookers use a spring valve with several pressure settings rather than a weight on the cover. A certain pressure cooker has a volume of 6 L and an operating pressure of 75 kPa gage. Initially, it contains 1 kg of water. Heat is supplied to the pressure cooker at a rate of 500 W for 30 min after the operating pressure is reached. Assuming an atmospheric pressure of 100 kPa, determine the temperature at which cooking takes place.arrow_forwardPressure cookers, in general, maintain a gage pressure of 2 atm (or 3 atm absolute) inside. Therefore, pressure cookers cook at a temperature of about 133°C instead of 100°C, cutting the cooking time by as much as 70 percent while minimizing the loss of nutrients. The newer pressure cookers use a spring valve with several pressure settings rather than a weight on the cover. A certain pressure cooker has a volume of 6 L and an operating pressure of 75 kPa gage. Initially, it contains 1 kg of water. Heat is supplied to the pressure cooker at a rate of 500 W for 30 min after the operating pressure is reached. Assuming an atmospheric pressure of 100 kPa, determine the amount of water left in the pressure cooker at the end of the process.arrow_forward
- The tension in the walls of a horizontal 0.1 m diameter pipe under a static pressure of 10000 cm of water and submerged to a depth of 10 m in a fresh-water river is:arrow_forward6 -Problem II - A volume of compressed air storage tank is 0.3m3, and the air pressure P1 in the tank is 1MP.. When a part of compressed air is used, the pressure P2 of the air in the tank decreases to 0.9 MPa. Set the room temperature at 20 ° C, calculate the mass of original air ml and the mass of residual air m2. Air gas constant Rg = 287.1 J/ (kg - K)). 21arrow_forwarda) In a simple experiment, a solid with a diameter 3 mm and density 1500 kg/m3is released from a tall tower containing air at 25 oC and 1 atm. The inside diameter of the column is 0.15 m. The density of the air is assumed to be 1.2 kg/m3. List all your assumptions and estimate the minimum height of the tower to make sure the particle reaches its terminal velocity before it hits the bottom of the tower. b) In another experiment, 5 kg of the solid particles are to be fluidized with air at 1 atm and 25 oC. The diameter and density of the particles are 3 mm and 95 kg/m3, respectively.Using the Ergun Equation, determine whether the flow of the air is laminar orturbulent. List all your assumptions.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





