
HUMAN PHYSIOLOGY: AN INTEG ACCESS C
8th Edition
ISBN: 9780134714837
Author: Silverthorn
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.1, Problem 7CC
Two compartments are separated by a membrane that is permeable to water and urea but not to NaCl. Which way will water move when the following solutions are placed in the two compartments? (Hint: Watch the units!)
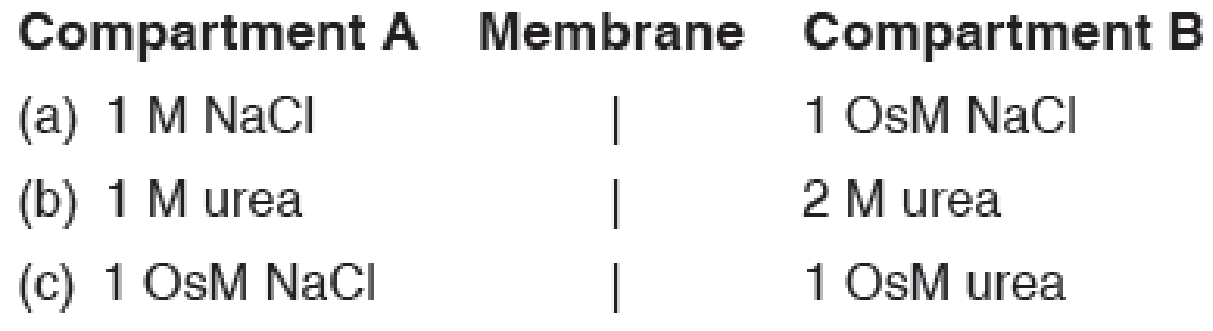
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The greatest buffering capacity at physiological pH would be provided by a protein rich in which of the following amino acids? Choose one from among the possible answers and explain.
Serine
Cysteine
Alanine
Histidine
What is the molar oconcentration of 30g KCl and 30g of CaCl2 added to 50 dL of DI water?
a.) what is the molar concerntration of Ca2+
b.) what is the molar concentration of K+
c.) what is the molar concentration of Cl-
d.) what is the osmolarity of this solution?
e.) what is the tonicity of this solution?
The simple form of |Hoff equation is:
II = [B]RT
In this equation the [B] is the molar concentration of solute. So:
n
m
[B] =
v MV
= cg /MA
Where c, the mass concentration of the solute is in the total volume of solution and M, is the molar mass of the solute. This equation can be
replaced in the previous one to get:
RT
II =
MA
In this equation molar mass of given solute can be detemined from the slope of the II vs Cz plot. This equation applies only to solutions that are
sufficiently dilute to behave as ideal-dilute solutions. In the case of non-ideal solutions, however, the extended formula is:
II = [B]RT{1+ k. [B] + n. [B]² + ...}
Biological macromolecules dissolve to produce solutions that are far from ideal, but we can still calculate the osmotic pressure by assuming that
the van't Hoff equation is only the first term of a lengthier expression:
II [B]RT(1+ b. [B])
II
= RT + bRT. [B]
[B]
II
= RT + bRT./M.
*/Ma
п
RT ÞRT
Ca
MA
MA
In this equation molar mass of given biomolecule can…
Chapter 5 Solutions
HUMAN PHYSIOLOGY: AN INTEG ACCESS C
Ch. 5.1 - If the 58-kg Reference Woman has total body water...Ch. 5.1 - A mother brings her baby to the emergency room...Ch. 5.1 - Prob. 6CCCh. 5.1 - Two compartments are separated by a membrane that...Ch. 5.1 - Prob. 8CCCh. 5.1 - Prob. 9CCCh. 5.1 - Prob. 10CCCh. 5.3 - If the distance over which a molecule must diffuse...Ch. 5.3 - Prob. 12CCCh. 5.3 - Which is more likely to cross a cell membrane by...
Ch. 5.3 - Prob. 14CCCh. 5.3 - Prob. 15CCCh. 5.3 - Prob. 16CCCh. 5.4 - Positively charged ions are called _____, and...Ch. 5.4 - Name four functions of membrane proteins.Ch. 5.4 - Prob. 19CCCh. 5.4 - Prob. 20CCCh. 5.4 - If a channel is lined with amino acids that have a...Ch. 5.4 - Prob. 22CCCh. 5.4 - Liver cells (hepatocytes) are able to convert...Ch. 5.4 - Prob. 24CCCh. 5.5 - What would you call a carrier that moves two...Ch. 5.5 - Prob. 26CCCh. 5.5 - Prob. 27CCCh. 5.5 - Name the two membrane protein families associated...Ch. 5.5 - Prob. 29CCCh. 5.6 - Prob. 30CCCh. 5.6 - Prob. 31CCCh. 5.6 - Prob. 32CCCh. 5.6 - Prob. 33CCCh. 5.7 - Prob. 34CCCh. 5 - Using what you learned about the naming...Ch. 5 - Prob. 2CCCh. 5 - Prob. 3CCCh. 5 - Prob. 1RQCh. 5 - Distinguish between active transport and passive...Ch. 5 - Which of the following processes are examples of...Ch. 5 - List four factors that increase the rate of...Ch. 5 - List the three physical methods by which materials...Ch. 5 - A cotransporter is a protein that moves more than...Ch. 5 - Prob. 7RQCh. 5 - Prob. 8RQCh. 5 - Prob. 9RQCh. 5 - What determines the osmolarity of a solution? In...Ch. 5 - Prob. 11RQCh. 5 - Prob. 12RQCh. 5 - Prob. 13RQCh. 5 - Prob. 14RQCh. 5 - The membrane potential at which the electrical...Ch. 5 - Prob. 16RQCh. 5 - Create a map of transport across cell membranes...Ch. 5 - Draw a large rectangle to represent the total body...Ch. 5 - What factors influence the rate of diffusion...Ch. 5 - Define the following terms and explain how they...Ch. 5 - Prob. 21RQCh. 5 - Prob. 22RQCh. 5 - Prob. 23RQCh. 5 - Prob. 24RQCh. 5 - Prob. 25RQCh. 5 - Prob. 26RQCh. 5 - The following terms have been applied to membrane...Ch. 5 - Prob. 28RQCh. 5 - NaCl is a nonpenetrating solute and urea is a...Ch. 5 - Prob. 30RQCh. 5 - Prob. 31RQCh. 5 - What is the osmolarity of half-normal saline (=...Ch. 5 - Prob. 33RQCh. 5 - Prob. 34RQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- An amino acid mixture consisting of phenylalanine, glycine, and glutamic acid is to be separated by HPLC. The stationary phase is aqueous and the mobile phase is a solvent less polar than water. Which of these amino acids will move the fastest? Which one will move the slowest?arrow_forwardIs lysine a suitable buffer at acidic, neutral, or basic pH range, and why?arrow_forwardwhy does water move inward in a hypotonic solution?arrow_forward
- hosphate buffered saline (PBS) is a buffer solution commonly used in biological The buffer helps to maintain a constant pH. The osmolality and ion concentrations of the solution usually match those of the human body. You need to prepare a stock solution at pH 7.00 with NaH2PO4 and Na2HPO4 (pKa =7.21). What would be the respective concentration of these substances if you wish to obtain the final phosphate concentration [HPO4 −2] + [H2PO4 −] = 0.3 M? What will be the pH of the solution after adding 0.18 L of 0.2 M NaOH? What is the ratio of HPO4−2 of H2PO4 – is part b, and what is the significance? NON GRADED PRACTICE QUESTION FOR BIOCHEMarrow_forwardMannitol, a carbohydrate, is supplied as a 25% (w/v) solution. This hypertonic solution is given to patients who have sustained a head injury with associated brain swelling. (a) What volume should be given to provide a dose of 70. g? (b) How does the hypertonic mannitol benefi t brain swelling?arrow_forwardWhen 2.43 g of a nonelectrolyte solute is dissolved in water to make 435 mL of solution at 25 °C, the solution exerts an osmotic pressure of 895 torr. What is the molar concentration of the solution? concentration: 0.045 M Incorrect How many moles of solute are in the solution? moles of solute: 0.034 mol Incorrect What is the molar mass of the solute? molar mass: 69.12 g/mol Incorrectarrow_forward
- Please note when values are from an external source/table. List any assumptions.arrow_forwardYou are trying to separate a mixture of AMP, ADP, and ATP using ion exchange chromatography. Which of the following is NOT an appropriate reagent for this experiment? You can not separate nucleotides using ion exchange chromatography. A positively charged column Protonated buffer 0.05 M NaCl buffer (constant concentration)arrow_forwardthe characteristics of water insoluble substances that is most important in governing its diffusibility through a cell membrane is its?arrow_forward
- Define the following:- pH- Buffer- pKaarrow_forwardIf you added three proteins (Protein A, pI 5.0; Protein B, pI 6.0; Protein C, pI 7.0) to an anion exchange column equilibrated with bicine buffer, pH 8.5, in what order would these protein be expected to elute as the column was loaded, washed, and then increasing amounts of NaCl were added?arrow_forwardA 1.143 g sample contains only vitamin C (C,H, 0,) and sucralose (C,H,Cl, O,). When the sample is dissolved in water to a total volume of 31.7 mL, the osmotic pressure of the solution is 3.91 atm at 285 K. What is the mass percent of vitamin C and sucralose in the sample? vitamin C: sucralose: %arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

The Cell Membrane; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=AsffT7XIXbA;License: Standard youtube license