
Concept explainers
(a)
Interpretation: The increasing stability of carbocation is to be interpreted.
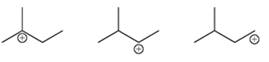
Concept introduction: Intermediates like carbocation can rearrange to give more stable carbocation. A primary carbocation carries a positive charge on the primary carbon atom whereas a secondary carbocation has a positive charge on the secondary carbon atom. Due to more hyperconjugation and inductive effect, the order of stability is:
Tertiary >Secondary >Primary
(b)
Interpretation: The increasing stability of carbocation is to be interpreted.
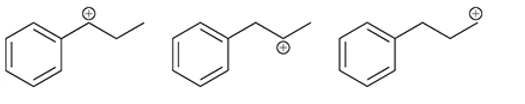
Concept introduction: Intermediates like carbocation can rearrange to give more stable carbocation. A primary carbocation carries a positive charge on the primary carbon atom whereas a secondary carbocation has a positive charge on the secondary carbon atom. Due to more hyperconjugation and inductive effect, the order of stability is:
Tertiary >Secondary >Primary
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
ORGANIC CHEMISTRY
- Rank the following carbocations in order of decreasing stability (from the most stable to the least stable): O A B C D Earrow_forwardGive IUPAC name for the following compounds: (Specify stereochemistry when shown in the diagram.) Ph Hi CH3CH2 1st compound: 2nd compound: H HO OH "H H CHCH₂CH₂CH3arrow_forwardRank the carbocation compounds below from most stable to least stable.arrow_forward
- When (R)-3-bromo-2,3-dimethylpentane is treated with NaOH, four different alkenes are formed. Draw all four products and rank them in order of stability.arrow_forwardRank the carbocations from most to least stable: O A B N сarrow_forwardRank the following alkenes in order of increasing stability: A вarrow_forward
- Rank the following alkenes in order of increasing stability:arrow_forwardLabel each pair of alkenes as constitutional isomers or stereoisomers. Select the single best answer for each part below. Part 1 of 3 H₂C=CHCH₂CH₂CH₂CH₂CH₂CH3 and H₂CH₂CHC=CHCH₂CH₂CH₂CH₂ O stereoisomers O constitutional isomers Part 2 of 3 H₂C H Part 3 of 3 O constitutional isomers O stereoisomers H₂C H CH₂CH3 H₂C H X CH₂CH₂CH₂CH₂ H O constitutional isomers O stereoisomers X CH₂ H X H₂C 5 CH₂CH₂CH₂CH₂ H 00 ← B 00 Ar !arrow_forwardRank the following carbocations in order of decreasing stability a A, B, C B, A, C OC, B, A B, C, A С, А, В A, C, B b C +arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





