
Concept explainers
(a)
Interpretation: The missing lone pairs and curved arrows are to be interpreted for the accomplishment of the given transformation:
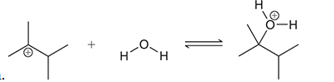
Concept introduction: One or more reactant molecules are changed into product molecules during a
The shifting of electron pairs depends on the electrophile and nucleophilic nature of the bonded atoms.
(b)
Interpretation: The missing lone pairs and curved arrows are to be interpreted for the accomplishment of the given transformation:
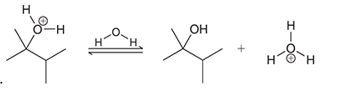
Concept introduction: One or more reactant molecules are changed into product molecules during a chemical reaction. It entails the movement of lone pairs on the connected atoms as well as bonding electrons.
The shifting of electron pairs depended on the electrophile and nucleophilic nature of the bonded atoms.
(c)
Interpretation: The missing lone pairs and curved arrows are to be interpreted for the accomplishment of the given transformation:
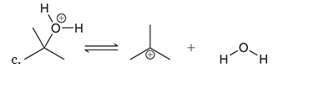
Concept introduction: One or more reactant molecules are changed into product molecules during a chemical reaction. It entails the movement of lone pairs on the connected atoms as well as bonding electrons.
The shifting of electron pairs depended on the electrophile and nucleophilic nature of the bonded atoms.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
ORGANIC CHEMISTRY
- . Use the curly arrow formalism to illustrate how the transformations shown below might occur. CH₂ (+) A CH₂arrow_forwardDraw the Newman projection so that it corresponds to the molecule and conformation shown when viewed down the red bond in the direction of the arrow. н с H H CH,arrow_forward(1) According to the Hammond Postulate, the transition state of this reaction will look like A) reactants B) intermediates C) products D) Neither the reactant or the products are good models for the structure of this transition state.arrow_forward
- How many degrees of unsaturation do the following molecules have? b)arrow_forwardDraw all four unique conformations (using Newman projections) for the molecule shown below, using the templates that have been provided. The arrow indicated the bond you will sight down in order to draw your Newman projections. Label these conformations as being either staggered or eclipsed. Also indicate which of these conformations is the most stable.arrow_forwardPlease answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Long and detailed explanations are not needed. Keep it short, brief, and direct because I only need the answers as soon as possible. The energy diagram below shows the conformational changes during rotation around the C–C bond in compound a. Which letter(s) on the graph correspond(s) to the Newman Projection shown to the right of compound a?arrow_forward
- Draw the Newman projection so that it corresponds to the molecule and conformation shown when viewed down the red bond in the direction of the red arrow. Your projection should be oriented as shown by the arrow marked up. So the CH2SH group on the front carbon should be above the H and H3C groups, no matter which template you usearrow_forwardFor the following Newman Projection, determine whether it is in the lowest energy conformation as drawn. Then, fill in the Newman Projection for the lowest energy conformation. You may use the table of A-values provided in the instructions to determine the correct answer. 2. H 3. CH3 (Me) Is this the lowest energy conformation? No For each number on the partially filled Newman Projection above, select the group that would make the lowest energy conformation. 1. CH2CH3 (Et) 4. H 5. CH(CH3)2 (i-prop) 5. 47 2 CH3 .3arrow_forwardDraw the curved arrows that accomplish each of the following transformations. H by H CH3 Lave CH3 + CH3 H3C H Edit Drawing O-H H3C H3C +H CH3 CH3arrow_forward
- Which of the following projections would the most stable? I believe that it would be the 6th because out of all 6 groups, the methyl groups are the furthest distance away from each other in the 6th. When they are in close proximity, the front and back methyl groups’ electron orbitals repel each other, which generates potential energy and makes the conformation unstable.arrow_forwardDraw 2,4-dimethylhexane as a bond-line structure. Then, looking down the C3 – C4 bond, draw a Newman projection representation of the 1) lowest energy (most stable) and 2) highest energy (least stable) conformations.arrow_forward4. Draw the most stable conformation for each of the following substituted cyclohexanes; then, in each case, flip the ring and redraw the molecular in the higher energy chair conformation. Shown all conformation in Newman projections. Fill in hydrogens to indicate unsubstituted carbons. a) b) most stable conformation: higher energy conformation: á most stable conformation: higher energy conformation: 3 Daarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





