
(a)
Interpretation: The arrow pushing method is to be interpreted for the given transformation.
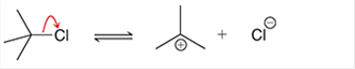
Concept introduction: The chemical transformation can occur with the hydride shift; methylene shifts or methyl shifts etc.
Each transformation can be shown by the arrow pushing method in which the curved arrows show the movement of electrons or protons. The chemical transformation leads to the formation of intermediates.
(b)
Interpretation: The arrow pushing method is to be interpreted for the given transformation.
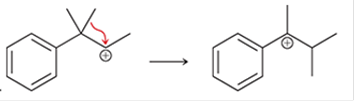
Concept introduction: The chemical transformation can occur with the hydride shift; methylene shifts or methyl shifts etc.
Each transformation can be shown by the arrow pushing method in which the curved arrows show the movement of electrons or protons. The chemical transformation leads to the formation of intermediates.
(c)
Interpretation: The arrow pushing method is to be interpreted for the given transformation.
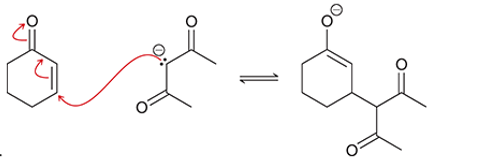
Concept introduction: The chemical transformation can occur with the hydride shift; methylene shifts or methyl shifts etc.
Each transformation can be shown by the arrow pushing method in which the curved arrows show the movement of electrons or protons. The chemical transformation leads to the formation of intermediates.
(d)
Interpretation: The arrow pushing method is to be interpreted for the given transformation.
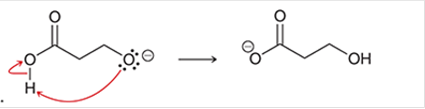
Concept introduction: The chemical transformation can occur with the hydride shift; methylene shifts or methyl shifts etc.
Each transformation can be shown by the arrow pushing method in which the curved arrows show the movement of electrons or protons. The chemical transformation leads to the formation of intermediates.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
ORGANIC CHEMISTRY
- Draw all the intermediates formed for the reactions shown. (Hint: There are five intermediates for the first reaction and two for the second reaction, and the final product shown is not counted, of course.) LOH OH 0 HO H+ заarrow_forwardPredict the major reaction pathway that will occur under these conditions. SN1 SN2 E1 E2 Br NaOAc ACOHarrow_forwardTell which of these transformations are oxidations and which are reductions based on whether there is addition or removal of O or H.arrow_forward
- 4) Multistriatin, one of the pheremones of the elm bark beetle, is a volatile compound released by a virgin female beetle when she has found a good food source - an elm tree. Male beetles, which carry the Dutch elm disease fungus, are attracted by the pheromone; the tree becomes infected with the fungus and soon dies. Multistriatin is used to trap the beetles, but the amount in beetles is so miniscule that the compound must be synthesized. The following synthesis of multistriatin uses only chemistry that we have seen in Chem 348. HO. НО Br type O type. H type H H CH3 H HO HO type O H CH3 "Η OH type H hint: anhydrous cond CH3 H = H type type HO CH3 H H type CH 3 H Multistriatin Multistriatinarrow_forwardFor each reaction in question 20, sketch a reaction coordinate energy diagram indicating the starting materials, transition states and any intermediates, if present, in the reaction. Don't worry about the absolute energy of starting materials and products but you can assume that the product is more stable than the starting materials.arrow_forwardList the appropriate reagents that can be used in places with questions in the following series of reactionsarrow_forward
- When propene reacts with gaseous hydrogen bromide, HBr, two products, 1-bromopropane and 2-bromopropane are formed. The reaction is a two-step process in which the electrophilic attack occurs in the first step. Identify the electrophile in this reaction Draw a diagram showing the first step of the reaction that leads to the production of 2-bromopropane.arrow_forwardThe number of different monochlorinated constitutional isomers produced when each of the following undergo a uv light catalysed reaction with chlorine: Compound i: 2 Compound: ii: 1 Compound i: 3 Compound: ii: 4 Compound i: 4 Compound: ii: 2 Compound i: 5 Compound: ii: 1 Compound i: 6 Compound: ii: 3arrow_forwardFor the following reaction there are three (3) possible products. Two of the products are expected to be minor and one product is expected to be a major product. Br H₂O Name and draw out the step-by-step mechanisms by which each of the products is formed. Briefly explain why one of the three products is expected to be a major product.arrow_forward

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning




