
Concept explainers
(a)
Interpretation:
The Lewis structure of
Concept Introduction:
A Lewis structure is also known as Lewis dot diagrams or electron dot structures. The bond between atoms and lone pairs of electrons that is present in the molecule. Lewis structure represents each atom and their position in structure using the chemical symbol. Excess electrons forms the lone pair are given by pair of dots, and are located next to the atom.
The formula for the formal charge can be written as
(a)
Explanation of Solution
The correct Lewis structure of the
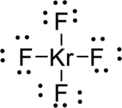
Draw a chart:
The total number of valence electron is
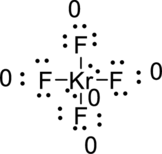
(b)
Interpretation:
The Lewis structure for
Concept Introduction:
Refer Part (a).
(b)
Explanation of Solution
The correct possible Lewis structure for
First structure:
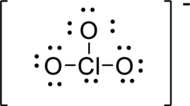
Draw a chart:
The total number of valence electron is
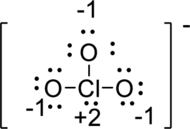
To make double bond between chlorine and oxygen against the octet rule and then reduced the formal charge to zero.
Draw a chart:
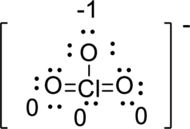
(c)
Interpretation:
The Lewis structure of
Concept Introduction:
Refer Part (a).
(c)
Explanation of Solution
The correct Lewis structure of the
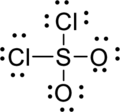
Draw a chart:
The total number of valence electron is
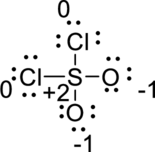
To make double bond between sulfur and oxygen against the octet rule and then reduced the formal charge to zero.
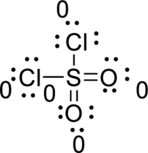
Hence, the formal charge of the
Want to see more full solutions like this?
Chapter 6 Solutions
Chemistry: The Molecular Science
- Write the correct Lewis structure and assign a formal charge to each atom in fulminate ion, CNO.arrow_forwardWrite all resonance structures of chlorobenzene, C6H5Cl, a molecule with the same cyclic structure as benzene. In all structures, keep the CCl bond as a single bond. Which resonance structures are the most important?arrow_forwardDraw Lewis structures for these ions and show which atom (or atoms) in each bears the formal charge. Q.) OH-arrow_forward
- What is the formal charge on each atom in HNO3 and the Lewis structure for it?arrow_forwardWrite a Lewis structure for HC2─ and assign any formal charges to the correct atom.arrow_forwardDraw three resonance structures for carbonate ion, CO32-, and assign formal charges on all the atoms.arrow_forward
- The cyanate ion, NCO– , has three (3) possible Lewis structures. (a) Draw these three structures and assign formal charges in each. (b) Which Lewis structure is dominant?arrow_forwardDraw a Lewis structure for H3C—NH2. Based on this Lewis structure, what is the value for the formal charge on the nitrogen atom?arrow_forwardDraw the Lewis structure with lowest formal charges, anddetermine the charge of each atom in (a) BF₄⁻; (b) ClNO.arrow_forward
- The thiocyanate ion , NCS- , has three possible Lewis structures.a. Draw these three structures and assign formal charges in each.b. Which Lewis structure is dominant?arrow_forwardThe cyanate ion, NCO-, has three possible Lewis structures.(a) Draw these three structures and assign formal charges ineach. (b) Which Lewis structure is dominant?arrow_forwardWrite the Lewis structures for CH2N2, including all resonance forms, and show formal charges.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

