
ORG.CHEMISTRY W/ACCESS+MODEL KIT PKG
5th Edition
ISBN: 9781260701128
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 6.3P
By taking into account electronegativity differences, draw the products formed by heterolysis of the carbon-heteroatom bond in each molecule. Classify the organic reactive intermediate as a carbocation or a carbanion.
a.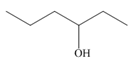 b.
b. 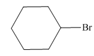 c.
c. 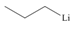
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
What are the MAJOR organic product for each reaction??
Draw the product when A (TOP) and B (BOTTOM) react together:
(Ignore the SOCl2)
Draw the Organic products E and F and classify the chemical transformation
Chapter 6 Solutions
ORG.CHEMISTRY W/ACCESS+MODEL KIT PKG
Ch. 6 - Problem 6.1 Classify each transformation as...Ch. 6 - Prob. 6.2PCh. 6 - Problem 6.3 By taking into account...Ch. 6 - Problem 6.4 Use curved arrows to show the movement...Ch. 6 - Problem 6.5 Follow the curved arrows and draw the...Ch. 6 - Prob. 6.6PCh. 6 - Problem 6.7 Use the values in Table 6.2 to...Ch. 6 - Prob. 6.8PCh. 6 - aWhich Keq corresponds to a negative value of G,...Ch. 6 - Given each of the following values, is the...
Ch. 6 - Given each of the following values, is the...Ch. 6 - The equilibrium constant for the conversion of the...Ch. 6 - Prob. 6.13PCh. 6 - For a reaction with H=40kJ/mol, decide which of...Ch. 6 - For a reaction with H=20kJ/mol, decide which of...Ch. 6 - Draw an energy diagram for a reaction in which the...Ch. 6 - Prob. 6.17PCh. 6 - Prob. 6.18PCh. 6 - Problem 6.19 Consider the following energy...Ch. 6 - Draw an energy diagram for a two-step reaction,...Ch. 6 - Which value if any corresponds to a faster...Ch. 6 - Prob. 6.22PCh. 6 - Problem 6.23 For each rate equation, what effect...Ch. 6 - Prob. 6.24PCh. 6 - Identify the catalyst in each equation. a....Ch. 6 - Draw the products of homolysis or heterolysis of...Ch. 6 - Explain why the bond dissociation energy for bond...Ch. 6 - Classify each transformation as substitution,...Ch. 6 - Prob. 6.29PCh. 6 - 6.30 Draw the products of each reaction by...Ch. 6 - 6.31 (a) Add curved arrows for each step to show...Ch. 6 - Prob. 6.32PCh. 6 - Prob. 6.33PCh. 6 - Prob. 6.34PCh. 6 - Calculate H for each reaction. a HO+CH4CH3+H2O b...Ch. 6 - Homolysis of the indicated CH bond in propene...Ch. 6 - Prob. 6.37PCh. 6 - Prob. 6.38PCh. 6 - 6.39. a. Which value corresponds to a negative...Ch. 6 - Prob. 6.40PCh. 6 - For which of the following reaction is S a...Ch. 6 - Prob. 6.42PCh. 6 - Prob. 6.43PCh. 6 - 6.44 Consider the following reaction: .
Use curved...Ch. 6 - Prob. 6.45PCh. 6 - Draw an energy diagram for the Bronsted-Lowry...Ch. 6 - Prob. 6.47PCh. 6 - Indicate which factors affect the rate of a...Ch. 6 - Prob. 6.49PCh. 6 - 6.50 The conversion of acetyl chloride to methyl...Ch. 6 - Prob. 6.51PCh. 6 - Prob. 6.52PCh. 6 - The conversion of (CH3)3Cl to (CH3)2C=CH2 can...Ch. 6 - 6.54 Explain why is more acidic than , even...Ch. 6 - Prob. 6.55PCh. 6 - Prob. 6.56PCh. 6 - Prob. 6.57PCh. 6 - Although Keq of equation 1 in problem 6.57 does...Ch. 6 - Prob. 6.59P
Additional Science Textbook Solutions
Find more solutions based on key concepts
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living by Chemistry
Write the electron configurations far each of the following elements: (a) Sc. (b) Ti. (c) Cr. (d) Fe. (e) Ru
Chemistry by OpenStax (2015-05-04)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw structural formulas for the major products of each reaction. (a) (b)arrow_forwardProvide an appropriate alkyne starting material A and intermediate product B. Omit byproducts. The number of carbon atoms in the starting material should be the same as in the final product.arrow_forwardWrite a stepwise mechanism for the following reaction.arrow_forward
- Give one example of Williamson ether synthesis. Show the reactants materials and the product. Draw the reaction mechanism?arrow_forwardRefer to the reaction energy diagram to answer parts (a) through (e). (a) The rate-limiting step is: ______. (b) The transition state for the fastest step is: _____. (c) In the third step which intermediate structurally resembles the transition state according to the Hammond postulate? _____. (d) Which step no. is endergonic? _____. (e) The number of intermediates in the overall reaction is: _____.arrow_forwardWhen trichloroacetaldehyde is dissolved in water, almost all of it is converted to the hydrate. Chloral hydrate, the product of the reaction, is a sedative that can be lethal. A cocktail laced with it is known—in detective novels, at least—as a “Mickey Finn.” Explain why an aqueous solution of trichloroacetaldehyde is almost all hydrate.arrow_forward
- When trichloroacetaldehyde is dissolved in water, almost all of it is converted to the hydrate. Chloral hydrate, the product of the reaction, is a sedative that can be lethal. A cocktail laced with it is known—in detective novels,at least—as a “Mickey Finn.” Explain why an aqueous solution of trichloroacetaldehyde is almost all hydrate.arrow_forwardDefine Reaction of Acetylide Anions ?arrow_forwardConsider the conversion of alkyl halide A to ether B. a.Classify the conversion of A to B as substitution, elimination, or addition. b.The reaction rate depends on the concentration of A only. Write the rate equation for the reaction, and explain why the reaction mechanism must involve more than one step. c.Heterolysis of the polar bond in A forms a resonance-stabilized intermediate. Draw all reasonable resonance structures for this intermediate.arrow_forward
- By taking into account electronegativity differences, draw the products formed by heterolysis of the carbon–heteroatom bond in each molecule. Classify the organic reactive intermediate as a carbocation or a carbanion.arrow_forwardDraw a reaction coordinate diagram for the reaction. (Hint: An alkyl halide is more stable than an alkene.) Draw the structure of the intermediate/s and their location/s in the above reaction coordinate diagram.arrow_forwardExplain why pentane-2,4-dione forms two different alkylation products (A or B) when the number of equivalents of base is increased from one to two.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY