
Interpretation:
The compound formed between elements C (Rb) and D (N) needs to be determined.
Concept introduction:
Molecules are bound together by different types of bonds namely ionic and covalent. Ionic bonds are formed when there is complete transfer of electrons from one atom to another. These atoms either lose or gain electrons to become negatively or positively charged ions. The forces of attraction between these ions causes the ionic bond formation.
Answer to Problem 68A
The compound formed between elements C and D is
Explanation of Solution
The given periodic table wherein elements C and D are marked.
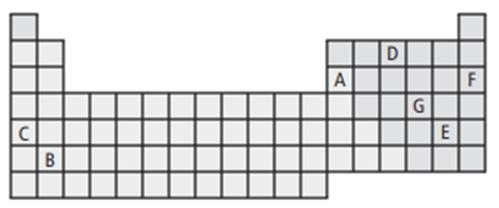
From the table, it can be seen that element C belongs to group 1 while element D belongs to group 15.
Element C is Rubidium and element D is Nitrogen.
Rubidium has
Nitrogen has atomic number of 7 with electronic configuration as
Hence, the compound formed between Rubidium and Nitrogen is
The compound between element C and element D is
Chapter 7 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Chemistry: The Central Science (14th Edition)
CHEMISTRY-TEXT
Essential Organic Chemistry (3rd Edition)
Chemistry: A Molecular Approach (4th Edition)
Organic Chemistry (8th Edition)
Chemistry: Structure and Properties (2nd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





