
Interpretation:
The charged particles of ionic compound is electrically neutral.
Concept introduction:
Chemical compounds are formed when the atoms, ions or molecules of two substances are attracted towards each other forming a
Answer to Problem 12SSC
The ionic compound has equal number of positively charged and negatively charged ions which is neutralized. Hence, is electrically neutral.
Explanation of Solution
A bond is formed when atom releases one or more electrons that is taken up by another atom. In due course, a negatively charge particle and positively charged particle is formed.
Ionic compounds are formed when negatively charged anion and positively charged cation are in close proximity. The number of positively charge particles are equal to number of negatively charged particles. These charges cancel each other and gets nullified. Hence, ionic compounds are said to be electrically neutral.
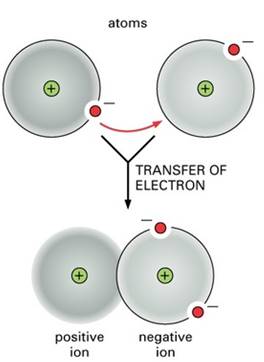
Figure 1
The ions are formed from atoms which either lose or gain electrons giving them a net positive or negative charge. Metals tend to lose electrons, forming positive ions or cations while non metals tend to gain electrons forming negative ions or anions.
The ionic compound has equal number of positively charged and negatively charged ions which is neutralized. Hence, is electrically neutral.
Chapter 7 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Chemistry: Structure and Properties
Organic Chemistry
Inorganic Chemistry
Introductory Chemistry (5th Edition) (Standalone Book)
Organic Chemistry (9th Edition)
Introductory Chemistry (6th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





