
Concept explainers
Interpretation:
The metallic bond needs to be explained.
Concept introduction:
The bonding between two or more atoms, molecules or ions results in
Answer to Problem 87A
The metallic bonds are those which are formed with the positive ions that are attracted to the delocalized electrons.
Explanation of Solution
Metallic bonds are formed as a result of overlapping of electronic orbitals. The electrons present in the electronic orbitals are shared between the atoms. The vacant p and d-orbitals in outer energy levels of a metal allows the free movement of electrons. In other words, this sea of electrons (negatively charged) attract positive kernels and is the main reason for conductivity, malleability and the metallic bonding in a metal.
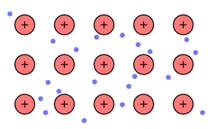
The positive ion (kernels) that flows in the negatively charged sea of electrons is the main reason of metallic bonding.
The metallic bonds are those which are formed with the positive ions that are attracted to the negatively charged delocalized electrons.
Chapter 7 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Organic Chemistry (9th Edition)
Organic Chemistry (8th Edition)
Inorganic Chemistry
CHEMISTRY-TEXT
Chemistry: A Molecular Approach
Chemistry: Structure and Properties (2nd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





