
Interpretation:
Formula for ionic compound formed by cesium and nitride needs to be determined.
Concept introduction:
Ionic compounds are formed as the result of formation of positive as well as negative ions. Electrons are basically transformed from one atom to other for formation of rare gas electron structure for each ion.
The atom that forms a positive ion will loses electrons to the atom that basically gains electrons for formation of a negative ion.
Answer to Problem 22PP
Formula is
Explanation of Solution
Formula for the ionic compounds formed by cesium and nitride is
The charge on Cs ion is +1 and that on N ion is -3 thus, to from neutral compound, 1 mol of N ion should react with 3 mol of Cs ion.
Chemical equation is represented as follows:
Here, 3 electrons from 3 Cs (1 electron each) are transferred to 1 N atom.
The electronic configuration is represented as follows:
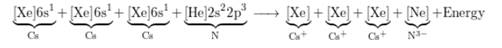
Therefore,
Chapter 7 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Introductory Chemistry (5th Edition) (Standalone Book)
Introductory Chemistry (6th Edition)
Essential Organic Chemistry (3rd Edition)
Organic Chemistry (9th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





