
ORGANIC CHEMISTRY 2 YEAR CONNECT ACCES
10th Edition
ISBN: 9781260025309
Author: Carey
Publisher: MCG/CREATE
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.4, Problem 8P
Give the IUPAC name of each of the compounds in Problem7.7, including E or Z as appropriate.
Determine the configuration of each of the following as Z or E as appropriate:
a) 
b) 
c) 
d) 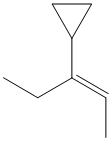
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Assign the absolute configuration of all molecules. Draw the most stable conformer in Newman projects with respect to bond C2-C3; for both a) and b). Draw Fisher projections for all molecules.
do in detail complete
Draw a newman projection of the given compound E viewed from C2-C3 oriented according to the first template. Then use the newman projection to demonstrate the most stable conformation around C2-C3 bond according to the second template
Calculate the number of degrees of unsaturation for each molecular formula:(a) C5H8O; (b) C6H11Cl; (c) C8H9N. Propose one possible structure for eachcompound.
Chapter 7 Solutions
ORGANIC CHEMISTRY 2 YEAR CONNECT ACCES
Ch. 7.1 - Name each of the following using IUPAC...Ch. 7.1 - Prob. 2PCh. 7.2 - How many carbon atoms are sp2-hybridized in the...Ch. 7.3 - Prob. 4PCh. 7.3 - Are cis-2-hexene and trans-3-hexene stereoisomers?...Ch. 7.4 - Prob. 6PCh. 7.4 - Prob. 7PCh. 7.4 - Give the IUPAC name of each of the compounds in...Ch. 7.5 - Arrange the following in order of increasing...Ch. 7.6 - Prob. 10P
Ch. 7.6 - Standard enthalpies of formation are known for all...Ch. 7.6 - Prob. 12PCh. 7.6 - Despite numerous attempts, the alkene...Ch. 7.6 - Write structural formulas for the six isomeric...Ch. 7.7 - Place a double bond in the carbon skeleton shown...Ch. 7.9 - Identify the alkene obtained on dehydration of...Ch. 7.10 - Prob. 17PCh. 7.11 - Prob. 18PCh. 7.12 - Prob. 19PCh. 7.13 - The alkene mixture obtained on dehydration of...Ch. 7.14 - Write the structures of all the alkenes that can...Ch. 7.14 - Write structural formulas for all the alkenes that...Ch. 7.15 - A study of the hydrolysis behavior of...Ch. 7.15 - Use curved arrows to illustrate the electron flow...Ch. 7.15 - Predict the major product of the reaction shown.Ch. 7.16 - Prob. 26PCh. 7.17 - Prob. 27PCh. 7.18 - Prob. 28PCh. 7.19 - Predict the major organic product of each of the...Ch. 7.19 - A standard method for the synthesis of ethers is...Ch. 7 - Write structural formulas for each of the...Ch. 7 - Prob. 32PCh. 7 - Give an IUPAC name for each of the following...Ch. 7 - A hydrocarbon isolated from fish oil and from...Ch. 7 - Prob. 35PCh. 7 - Prob. 36PCh. 7 - Prob. 37PCh. 7 - Prob. 38PCh. 7 - Choose the more stable alkene in each of the...Ch. 7 - Suggest an explanation for the fact that...Ch. 7 - Prob. 41PCh. 7 - Write structural formulas for all the alkene...Ch. 7 - Prob. 43PCh. 7 - Prob. 44PCh. 7 - Predict the major organic product of each of the...Ch. 7 - Prob. 46PCh. 7 - Prob. 47PCh. 7 - The rate of the reaction In the first order in...Ch. 7 - Prob. 49PCh. 7 - Prob. 50PCh. 7 - You have available 2,2-dimethylcyclopentanol (A)...Ch. 7 - Prob. 52PCh. 7 - Prob. 53PCh. 7 - Prob. 54PCh. 7 - Acid-catalyzed dehydration of...Ch. 7 - The ratio of elimination to substitution is...Ch. 7 - Prob. 57PCh. 7 - Prob. 58DSPCh. 7 - Prob. 59DSPCh. 7 - Prob. 60DSPCh. 7 - Prob. 61DSPCh. 7 - A Mechanistic Preview of Addition Reactions The...Ch. 7 - Prob. 63DSPCh. 7 - Prob. 64DSPCh. 7 - Prob. 65DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give the IUPAC name that corresponds to each compound. Indicate if E or Z conformation, cis or trans, R or S configuration where applicable.arrow_forwardAssign E or Z configuration to each of the following compounds: a. b. c. d.arrow_forwardCalculate the number of degrees of unsaturation for each molecular formula. a. C6H6 b. C40H56 c. C10H16O2 d. C8H9Br e. C8H9CIO f. CH711N g. C4H8BrN h. C10H18CINOarrow_forward
- Draw the most stable chair conformation of each of the following compounds. (a)cis-1,1,4 - trimethylcyclohexane (B) trans-1,1,3-trimethylcyclohexane (c) cis-1-fluro-4-ethylcyclohexane Please Give Step by Step Answer Otherwise i give DISLIKES !!arrow_forwardFor the compound below please choose the correct set of chair and flipped chair conformations:arrow_forwardcalculate the hydrogen deficiency of each compound c7h10cl2 c10h12n2o c4h3f7o draw a compound for #1arrow_forward
- Perform a confirmational analysis on cyclohexane derivative 1. Draw out both possible confórmeles and circle the preferred (lowest energy) conformer. Using A, U or G values estimate the energy difference ( Change in G) between the two confórmeles and predict the approximate ratio of the two confórmers at room temperature.arrow_forwardConsider the following compound represented in Newman projection:a) Draw this compound as shown in abbreviated structure (zig-zag with solid and hatched bevels):b) Using Newman's projections, draw the most stable AND least stable conformation while indicating whether it is an anti or left or eclipse conformation.arrow_forwardsight along the C2-C3 blond of 2-methylbutane: a)draw the newman projection of the most stable conformation b) draw the newman projection of the least stable conformation c)make a graph of the energy vs angle of rotation about the C2-C3 bond, and assign values to the y-axisarrow_forward
- For pentane draw Newman projections for the Syn-periplanar, conformation. the Anti- periplanar conformation and a Gauche conformation. Use C2 as the front carbon and C3 as the back carbon. Label each conformation, circle the highest energy conformation andunderline the lowest energy conformation.arrow_forwardConformer E is a (cis/trans) isomer. A conformational ring flip of conformer E will yield conformer F in which the substituent on carbon 1 will be (axial/equatorial/neither) and the substituent on carbon 3 will be (axial/equatorial/neither). Conformer (E/F) will be more stable.arrow_forwardDraw the three constitutional isomers having molecular formula C7H14 that contain a ve-membered ring and two methyl groups as substituents. For each constitutional isomer that can have cis and trans isomers, draw the two stereoisomers.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License