
a)
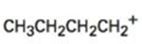
Interpretation:
The structure of more stable carbocation obtainable by the rearrangement of the n-butylcarbocation is to be proposed.
Concept introduction:
The stability of carbocations are in the order tertiary > secondary > primary. A carbocation will rearrange to a more stable carbocation either by a hydride shift or by an alkyl shift from adjacent carbon.
To propose:
The structure of more stable carbocation obtainable by the rearrangement of the carbocation given.
b)
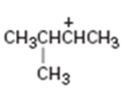
Interpretation:
The structure of more stable carbocation obtainable by the rearrangement of the carbocation given is to be proposed.
Concept introduction:
The stability of carbocations are in the order tertiary > secondary > primary. A carbocation can rearrange to a more stable carbocation either by a hydride shift or by an alkyl shift from adjacent carbon.
To propose:
The structure of more stable carbocation obtainable by the rearrangement of the carbocation given.
c)
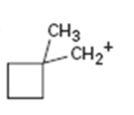
Interpretation:
The structure of more stable carbocation obtainable by the rearrangement of the carbocation given is to be proposed.
Concept introduction:
The stability of carbocations are in the order tertiary > secondary > primary. A carbocation will rearrange to a more stable carbocation either by a hydride shift or by an alkyl shift from adjacent carbon.
To propose:
The structure of more stable carbocation obtainable by the rearrangement of the carbocation given.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
- Provide the major product of each of the following reactions: What do all these reactions have in common? How do all reactions differ?arrow_forwardTwo of the carbocations are prone to rearrangement. Show how they might rearrange to more stable carbocationsarrow_forwardFor each of the following draw the major product and a mechanism to account for its formation. Be sure to include stereochemistry (if appropriate), arrows to show the movement of electrons and all formal charges.arrow_forward
- Which of the following carbocations would you expect to rearrange?arrow_forwardDraw the organic product for each of the below rearrangements. Be sure to include all nonzero formal charges.arrow_forwardFor the addition reaction below, determine the three possible products and show the FULL electron-pushing mechanism and ALL intermediates (including lone pairs and formal charges) to depicts how each one of the three products is obtained. Don't forget to explicitly show how the hydride shift happens to obtain the major product.arrow_forward
- Which of the following carbocations is(are) likely to rearrange? a. I b. II c. III d. II and III e. I and IIarrow_forwardFor each of the following reactions, state the mechanism the reaction will follow (E1,E2, SN1, SN2, etc.) and givethe major product(s) of the reaction.arrow_forwardFor each of the following reactions, state the mechanism the reaction (E1, E2, SN1, SN2, etc.) will follow and givethe major product(s) of the reaction.arrow_forward
- What is the complete mechanism using curved arrow formalism of the two products shown below? Explain why one is major and the other is minor product formation.arrow_forwardpredict the major products of the followijg reactionsarrow_forwardThe above reaction proceeds to yield only a single product. Draw the structure(s) of the carbocation intermediate, including resonance contributors, to show why this is the case.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

