
Concept explainers
Complete the following concept map that summarizes some of the key ideas about energy.
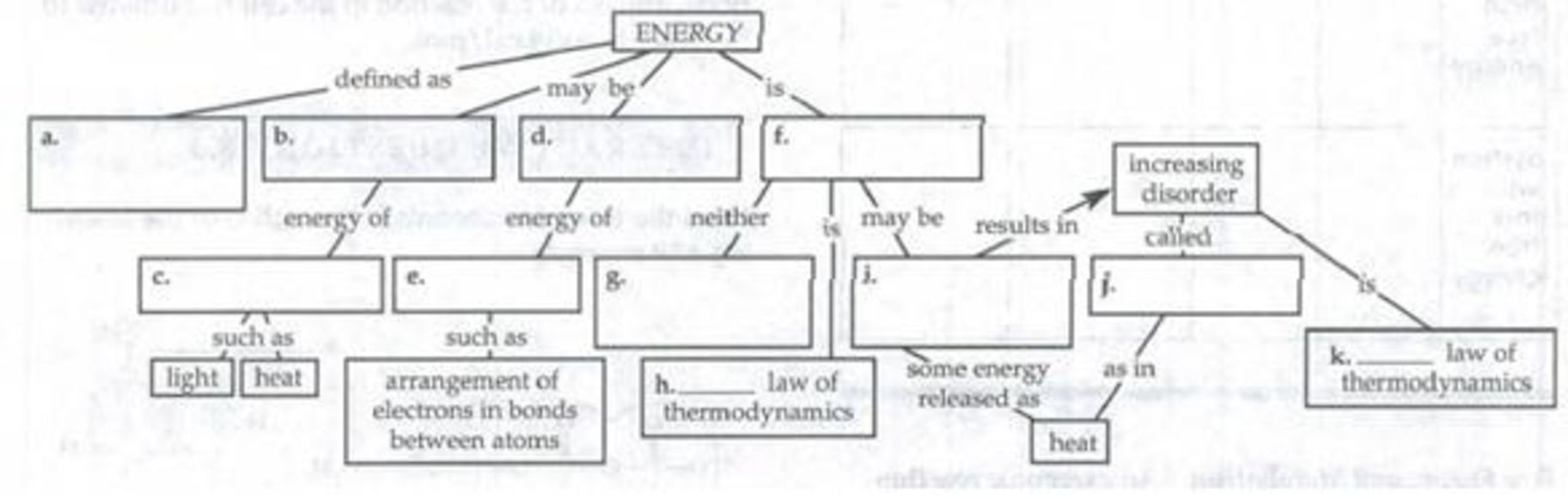
To complete: The concept map that summarizes some of the key ideas about energy.
Introduction: Thermodynamics is the study of energy and its transformation that drives all activities of the universe. The system that does not exchange energy with its surroundings is known as a closed system. The system that exchanges energy with its surroundings is known as an open system. There are two laws of thermodynamics, namely the first law and the second law.
Answer to Problem 1IQ
Pictorial representation: Fig.1 shows the concept map that summarizes some of the key ideas about energy.
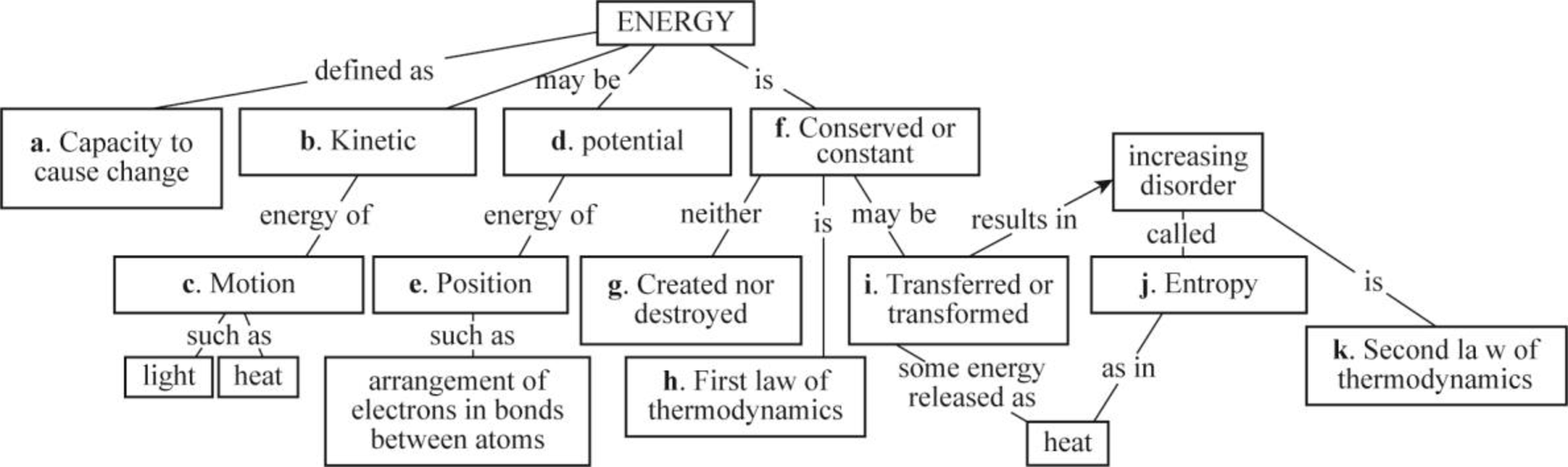
Fig.1: Concept map that summarizes some of the key ideas about energy.
Explanation of Solution
Energy is necessary for all living organisms to carry out their life processes. Living cells obtain energy in different forms, and they also have the mechanisms to convert the energy from one form to different forms. Energy is defined as the capability to do work. When work is done, energy is transferred from one form to another form, or between systems. Energy is generally expressed in units of work that is kilojoules (kJ). Energy can also be expressed in units of heat energy that is kilocalories (kcal).
Potential energy and kinetic energy are the two forms of energy. Potential is the energy of position or stored energy such as the arrangement of electrons in bonds between atoms. Kinetic energy is the energy of motion such as light and heat. A person pulling an arrow back on a bow and holding a baseball in the backswing are examples of potential energy. When the bowstring pushes the arrow toward its target and throwing a baseball are examples of kinetic energy.
Energy cannot be created and destroyed, but it can be converted from one form to another. It can tend to spread or disperse until no part of the system has more than one part. Entropy is a measure of how much energy a particular system has dispersed. The entropy of the system increases on heat dispersal, as molecule tends to disperse. This is a measure of randomness.
The first law of thermodynamics states that “energy cannot be created or destroyed, but it can be transformed from one form to another form”. This law explains that organisms cannot produce energy; however, they constantly capture energy from their surrounding as open systems and covert this energy from one form to another form for biological work.
The second law of thermodynamics states that “no energy transfer is 100% efficient; some energy is dispersed as heat”. According to this law, entropy in the universe is continuously increasing. As the organisms are open systems, they maintain their ordered states over time with a constant energy input from their surroundings.
Want to see more full solutions like this?
Chapter 8 Solutions
Biology - Study Guide
- Which of the following statements is not correct? a. Energy cannot be created or destroyed. b. Energy cannot change from one form to another. c. Energy tends to disperse spontaneously. d. Energy can be transferred.arrow_forwardselect which ones are examples of potential energyarrow_forwardUse the following graph to diagram the energetics of a chemical reaction, with and without an enzyme. Be sure to position reactants and products at appropriate points and to indicate the stages in the reaction and the energy levels.arrow_forward
- What is the minimum amount of energy required to start a chemical reaction? A. Activation energy B. Net energy input C. Potential energy D. Net energy outputarrow_forwardWhich are the several forms of energy important in biological systems.arrow_forwardWRITE A DESCRIPTION OF WHAT ENERGY TRANSFORMATIONS ARE HAPPENING AND WHY 1. BATTERY ➡ FLASHLIGHT 2. GASOLINE ➡ CARarrow_forward
- Choose the best answer for the following question? The sum of all the chemical reactions in a cell is calleda. free energy.b. entropy.c. metabolism.d. oxidation-reduction reactions.arrow_forwardWhich statement about energy is not correct? Energy is the ability to do work. A large ball on top of a hill has kinetic energy. The biological world gets its energy from the sun. Chemical bonds store potential energy. All forms of energy can be converted to heat.arrow_forward___________________ are molecules that influence the speed of chemical reactions, and are not consumed in the process.arrow_forward
- Define the following terms: a. thermodynamics b. bioenergetics c. entropy d. enthalpy e. free energyarrow_forwardSelect the best answer or answers from the choices given: Which of the following forms of energy is the stimulus for vision? (a) chemical, (b) electrical, (c) mechanical, (d) radiant.arrow_forwardStudy of energy is calledarrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning




