
Biology - Study Guide
8th Edition
ISBN: 9780321501561
Author: Martha R. Taylor
Publisher: PEARSON EDUCATION (COLLEGE)
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 2IQ
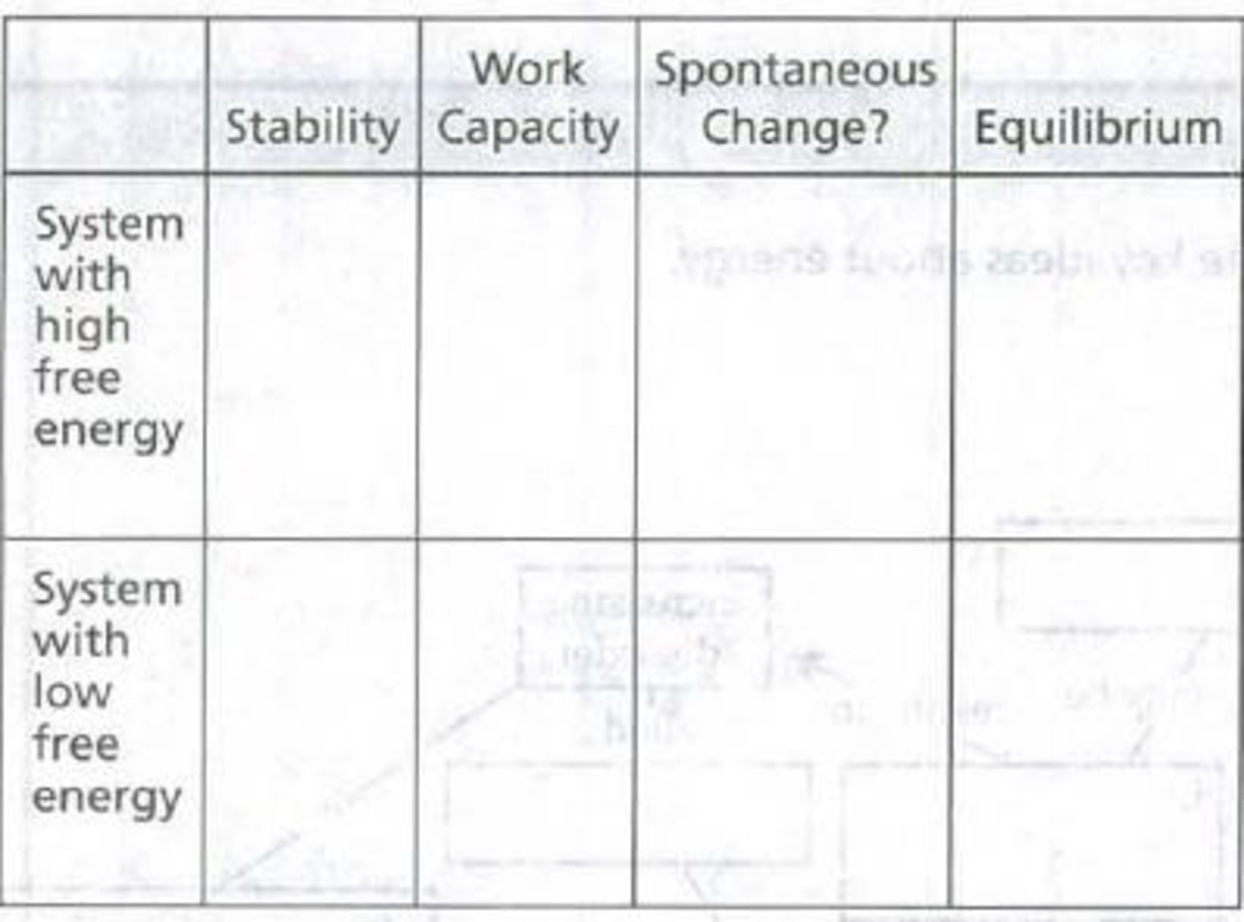
Complete the following table to indicate how the free energy of a system (or a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Define chemical equilibrium for reversible reactions.
Using collision theory, indicate which of the following statements regarding physical nature of the reactants is true:
a) physical nature of reactants
The physical nature of reactants does not influence the rate of reaction because reactions happen only on the molecular level.
Solids, liquids or gases all influence the rate of reaction the same because they are all in the physical state.
Solid state reactants react faster if the particle sizes are larger because reactions occur at the boundary surface with direct contact.
Gaseous-state reactions are faster than liquid-state or solid-state reactions because collisions between reactants are more frequent.
Use the following graph to diagram the energetics of a chemical reaction, with and without an enzyme. Be sure to position reactants and products at appropriate points and to indicate the stages in the reaction and the energy levels.
Chapter 8 Solutions
Biology - Study Guide
Ch. 8 - Complete the following concept map that summarizes...Ch. 8 - Complete the following table to indicate how the...Ch. 8 - Develop a concept map on free energy and G. The...Ch. 8 - Prob. 4IQCh. 8 - Prob. 5IQCh. 8 - In the following graph of an exergonic reaction...Ch. 8 - In the following diagram of a catalytic cycle,...Ch. 8 - Return to the diagram in Interactive Question 3.7,...Ch. 8 - Both ATP and ADP serve as regulators of enzyme...Ch. 8 - What is the relationship between the concept of...
Ch. 8 - What role do enzymes play in metabolism?Ch. 8 - ________ the totality of an organisms chemical...Ch. 8 - _______ pathways that use energy to synthesize...Ch. 8 - Prob. 3TYKFCh. 8 - _______ the most random form of energyCh. 8 - _______ term for the measure of disorder or...Ch. 8 - Prob. 6TYKFCh. 8 - _______ inhibitors that decrease an enzymes...Ch. 8 - Prob. 8TYKFCh. 8 - Prob. 9TYKFCh. 8 - Prob. 10TYKFCh. 8 - Catabolic and anabolic pathways are often coupled...Ch. 8 - Which statement most closely reflects the first...Ch. 8 - When a cell breaks down glucose, only about 34% of...Ch. 8 - Prob. 4TYKCh. 8 - Prob. 5TYKCh. 8 - Prob. 6TYKCh. 8 - One way in which a cell maintains metabolic...Ch. 8 - Prob. 8TYKCh. 8 - Prob. 9TYKCh. 8 - What is meant by an induced fit? a. The binding of...Ch. 8 - In an experiment, changing the pH from 7 to 6...Ch. 8 - Prob. 12TYKCh. 8 - Penicillin binds to the active site of an enzyme...Ch. 8 - Prob. 14TYKCh. 8 - Prob. 15TYKCh. 8 - Which line in the diagram indicates the G of the...Ch. 8 - Prob. 17TYKCh. 8 - Prob. 18TYKCh. 8 - Prob. 19TYKCh. 8 - Prob. 20TYK
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- List three of the five ways in which a nuclear reaction differs from a chemical reaction.arrow_forwardCompare the energy dynamics of a reaction at equilibrium with the dynamics of a reaction not at equilibrium.arrow_forwardDraw the structure of the chemical species represented by numbers for the following reactions.arrow_forward
- Sketch and label the various parts of transition state diagrams for a reaction with and without a catalyst.arrow_forwarddiscuss the following statement: “Whether the ΔG for a reaction is larger, smaller, or the same as ΔG° depends on the concentration of the compounds that participate in the reaction.”arrow_forwardDefine Kinetic energy and Potential energy.arrow_forward
- What is the most common form of kinetic energy that is released from chemical energy in chemical reactions?arrow_forwardA chemical species with an unpaired electron is called a_____________.arrow_forwardWrite an unbalanced equation, using the correct formulas for all reactants and products.arrow_forward
- The following equation, which represents the oxidative breakdown of glucose by body cells, is a reversible reaction. Glucose + oxygen S carbon dioxide + water + ATP (a) How can you indicate that the reaction is reversible? (b) Howcan you indicate that the reaction is in chemical equilibrium? (c) Define chemical equilibrium.arrow_forwardList the four factors that influence the rate of a chemical reaction and state whether increasing the factor will increase or decrease the rate of the reaction.arrow_forwardIn the following chemical reaction, which compounds are the products? Carbon dioxide + Water → Sugar + Oxygen A. Carbon dioxide and sugar B. Carbon dioxide and water C. Sugar and oxygen D. Water and oxygenarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning


Principles Of Radiographic Imaging: An Art And A ...
Health & Nutrition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Cengage Learning
Microbial Nutrition and Growth; Author: Scientist Cindy;https://www.youtube.com/watch?v=rK3UkyWjkl8;License: Standard YouTube License, CC-BY