
Concept explainers
(a)
Interpretation:
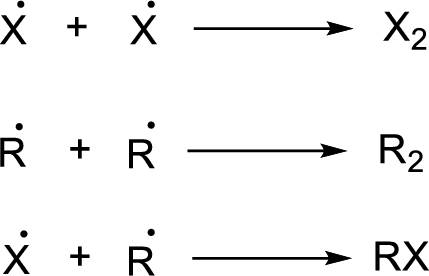
A second chain propagation step in the given reaction has to be proposed.
Concept introduction:
Halogenation of
Radical chain reaction:
Initiation reaction: 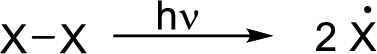
Chain propagation: 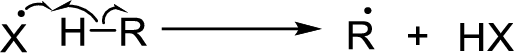
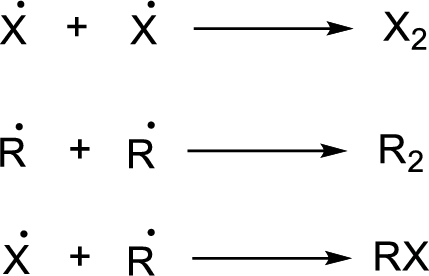
Chain termination:
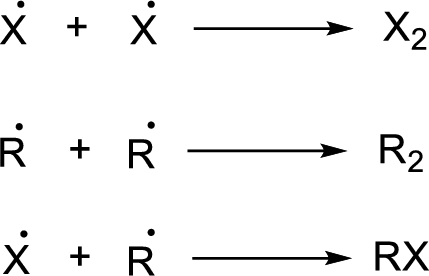
(b)
Interpretation:
Heat of the reaction,
Concept introduction:
Halogenation of alkanes: The replacement of one or more hydrogen atoms by halogen. When alkanes is heated or irradiated with the light of specific wavelength, the alkyl halide are formed by radical chain reaction.
Radical chain reaction:
Initiation reaction: 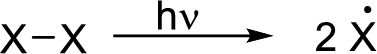
Chain propagation: 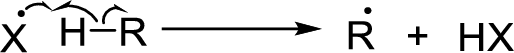
Chain termination:
It is a change in enthalpy of a homolysis reaction at absolute zero where a molecule is broken down into two free radicals.
(c)
Interpretation:
The energies and relative rates of the set chain propagation steps in section 8.5B with given reaction has to be compared.
Concept introduction:
Halogenation of alkanes: The replacement of one or more hydrogen atoms by halogen. When alkanes is heated or irradiated with the light of specific wavelength, the alkyl halide are formed by radical chain reaction.
Radical chain reaction:
Initiation reaction: 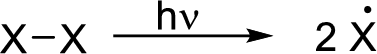
Chain propagation: 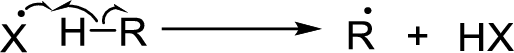
Chain termination:
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
- Fill the blank space. Compounds containing a phenol group may work as ANTIOXIDANTS to prevent free radical damage. This is accomplished when a free radical (or UV light) encounters a phenol group, turning the phenol group into a radical. However, contrary to typical radical behavior, the structure of the phenol radical can neutralize (or quench) the unpaired electron. Specifically, the phenol structure neutralizes (or quenches) the unpaired radical electron by doing the following: taking the electron and ---------. The correct name (or abbreviation) of an example compound containing a phenol group with antioxidant properties is: ---------.arrow_forwardA student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all the NBS has reacted. After a careful distillation, the product mixture contains two major products of formula C7H11Br. (a) Draw the resonance forms of the three possible allylic free radical intermediates.arrow_forwardWrite a step by step mechanism. Includes resonance structures that exist in the transition states.arrow_forward
- As we will learn, many antioxidants–compounds that prevent unwanted radical oxidation reactions from occurring–are phenols, compounds that contain an OH group bonded directly to a benzene ring.a.) Explain why homolysis of the O–H bond in phenol requiresconsiderably less energy than homolysis of the O–H bond in ethanol(362 kJ/mol vs. 438 kJ/mol).b.) Why is the C–O bond in phenol shorter than the C–O bond in ethanol?arrow_forwardThe above reaction involves heterolytic bond breakage of HBr a) Encircle the nucleophile (s) and electrophile (s) and explain why. b) Give the mechanism of the reaction by: ) Drawing the appropriate arrows to track the flow of electrons in Showing the species formed after bond breakage and bond formation with appropriate charges. c) Predict the final products.arrow_forwardThis molecule reacts with HBr to form an alkyl halide. Is this reaction Sn1 or Sn2 and explain your reasoning?arrow_forward
- a) Write the name of three steps of free-radical halogenation of methane?b) Give the mechanism of formation of dichloromethane from chloromethane?c) Explain why free-radical halogenation usually gives mixture of products.arrow_forwardWrite the complete reaction mechanism of this reaction including resonance structures of all the productsarrow_forwardWhen piperidine undergoes the series of reactions shown here, 1,4-pentadiene is obtained as the product. When the four different methyl- substituted piperidines undergo the same series of reactions, each forms a different diene: 1,5-hexadiene; 1,4-pentadiene; 2-methyl-1,4-pentadiene; and 3-methyl-1,4-pentadiene. Which methyl-substituted piperidine forms which diene?arrow_forward
- )a) The radical chlorination of pentane is a poor way to prepare 1-chloropentane but radical chlorination of neopentane, (CH).C, is a good way to prepare neopentyl chloride, (CH) CCH;CI. Explain. b) Show the mechanism for the radical chlorination of neopentane. Illustrate homolytic bond cleavage and bond formation.arrow_forwardb) Explain in detail what characteristics of the alkyl halide influence whether a mechanism will be SN1 or SN2. c) Explain in detail what characteristics of a nucleophile influence whether a reaction will be SN1 or SN2.arrow_forwardMechanism: Cl₂ → 2 CI* CI+ CH4 → HCI + CH3* CH3 + Cl₂ → CH3CI + CI CI+ CI → Cl₂ K₁ k₂ K3 K4 In the mechanism above comment on whether a third body could be used in certain steps and explain why. Write a brief note on when a third body may be necessary in radical reactions.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


