
Concept explainers
a)
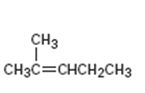
Interpretation:
The structure of the product obtained by the hydroboration-oxidation of 2-methyl-2-pentene is to be given.
Concept introduction:
The hydroboration reaction takes place with syn stereochemistry and results in a non-Markovnikov addition of water to the double bond in an
To give:
The structure of the product obtained by the hydroboration-oxidation of 2-methyl-2-pentene.
b)
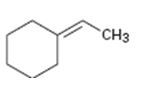
Interpretation:
The structure of the product obtained by the hydroboration-oxidation of the compound is to be given.
Concept introduction:
The hydroboration reaction takes place with syn stereochemistry and results in a non-Markovnikov addition of water to the double bond in the alkene. The resulting product has the hydroxyl group on the less highly substituted carbon.
To give:
The structure of the product obtained by the hydroboration-oxidation of the compound shown.
Trending nowThis is a popular solution!

Chapter 8 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Draw the structures of two alkenes that would react to form the haloalkane below upon addition of HBr. Your alkenes should be different regioisomers that yield the haloalkane as the major product without requiring rearrangement to occur.arrow_forwardYou are required to synthesize 2-bromopentane from the reaction between an alkene with HBr. Which alkene, 1-pentene or 2-pentene, should you react with HBr in order to get 2-bromopentane? Give an explanation.arrow_forwardCCH H20, H2SO4 H9SO4 CH3 Alkynes do not react directly with aqueous acid as do alkenes, but will do so in the presence of mercury(II) sulfate as a Lewis acid catalyst. The reaction occurs with Markovnikov regiochemistry, so the OH group adds to the more highly substituted carbon and the H adds to the less highly substituted carbon. The initial product of the reaction is a vinyl alcohol, also called an enol. The enol immediately rearranges to a more stable ketone via tautomerization. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions Hjö: -CH3 -CH3 H3O*arrow_forward
- Hydroxylation of E-2-pentene with osmium tetroxide yields a different product than the same reaction using the Z configuration. Draw the structures of the alkenes and show the stereochemistry for each product. Explain any differences between them.arrow_forwardAlkynes do not react directly with aqueous acid as do alkenes, but will do so in the presence of mercury(II) sulfate as a Lewis acid catalyst. The reaction occurs with Markovnikov regiochemistry, so the OH group adds to the more highly substituted carbon and the H adds to the less highly substituted carbon. The initial product of the reaction is a vinyl alcohol, also called an enol. The enol immediately rearranges to a more stable ketone via tautomerization. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions -X티 Hö: H-O -CH3 -CH3 H30*arrow_forwardC=CH H20, H2SO4 H9SO4 CH3 Alkynes do not react directly with aqueous acid as do alkenes, but will do so in the presence of mercury(II) sulfate as a Lewis acid catalyst. The reaction occurs with Markovnikov regiochemistry, so the OH group adds to the more highly substituted carbon and the H adds to the less highly substituted carbon. The initial product of the reaction is a vinyl alcohol, also called an enol. The enol immediately rearranges to a more stable ketone via tautomerization. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions H-OH HO: Hjö: C=CH c=CH Hö Hg Hgarrow_forward
- Suppose that the alkene in Figure 9 reacts with 1 mole of hydrogen chloride. Predict the structure of the main product of the reaction.arrow_forwardFor each of the following alkenes, assign the configuration of the double bond as either Z or E:arrow_forwardEthers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Draw the structure of the expected organic product of the reaction of iodomethane with the following alkoxide ion: CH3 H3C O Na You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Do not include counter-ions, e.g., Na", I, in your answer. орy вste ChemDoodlearrow_forward
- 22. Calculate the percent atom economy for the following hydroboration-oxidation reaction. This is another method of hydration of an alkene. Be careful with stoichiometry! You must multiply molecular weights by their stoichiometric coefficients. ОН 3 3 + BH3 +Na;BO3 + 3 H20 +3 H2O2 +3 NaOHarrow_forwardTwo substitution products result from the reaction between 3-chloro-3-methyl-1- butene with sodium acetate (CH3COO – Na +) in acetic acid under SN1. Identify the products.arrow_forwardWrite the structure of the major products in the following reactions and name themarrow_forward

 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

