
a)
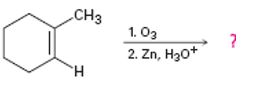
Interpretation:
The product of the reaction given showing both regiochemistry and stereochemistry is to be predicted.
Concept introduction:
Ozone adds to the double bond in
To give:
The product of the reaction given showing both regiochemistry and stereochemistry.
b)
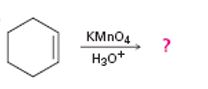
Interpretation:
The product of the reaction given showing both regiochemistry and stereochemistry is to be predicted.
Concept introduction:
Potassium permanganate in neutral or acidic solution cleaves alkenes to give carbonyl containing products. If hydrogens are present on the double bond
To predict:
The product of the reaction given showing both regiochemistry and stereochemistry.
c)
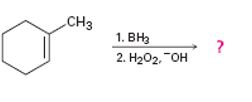
Interpretation:
The product of the reaction given showing both regiochemistry and stereochemistry is to be predicted.
Concept introduction:
The hydroboration reaction takes place with syn stereochemistry and results in a non-Markovnikov addition of water to the double bond in the alkene. The resulting product has the hydroxyl group on the less highly substituted carbon.
To predict:
The product of the reaction given showing both regiochemistry and stereochemistry.
d)
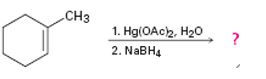
Interpretation:
The product of the reaction given showing both regiochemistry and stereochemistry is to be predicted.
Concept introduction:
In the oxymercuration-demercuration process, in the first step, the electrophilic addition of Hg2+ to the alkene gives a mercurinium ion. In the next step the mercurinium ion reacts with water to yield an organomercury product. In the last step, reaction with NaBH4 removes mercury to give a more highly substituted alcohol, corresponding to Markovnikov regiochemistry, as the product.
To predict:
The product of the reaction given showing both regiochemistry and stereochemistry
Trending nowThis is a popular solution!

Chapter 8 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Predict the major product of the following reactions. Indicate stereochemistry when relevant.arrow_forwardPredict the products of the following reactions. Show stereochemistry when applicable.arrow_forwardPredict the products of the following reactions. Show stereochemistry and regiochemistry of the product, where relevant.arrow_forward
- Predict the products and show the mechanisms for the following reactions. Please indicate the correct stereochemistry where necessary.arrow_forwardShow how the following starting materials are converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.arrow_forwardIdentify the following pericyclic reaction; explain the course and stereochemistry of the reaction.arrow_forward
- Predict the products and indicate the relative stereochemistry for the following transformations. When necessary indicate the major productarrow_forwardIdentify the following pericyclic reaction; explain the course, product distribution and stereochemistry of the reaction. Where the first product is produced 70% and the second product is produced 30%.arrow_forwardPredict the products for the following reactions, showing the complete mechanism and appropriate stereochemistry:arrow_forward
- Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardShow how the following starting material is converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.arrow_forwardPredict the products of the following reactions. Be sure to indicate stereochemistry when necessaryarrow_forward
