
Concept explainers
Interpretation:
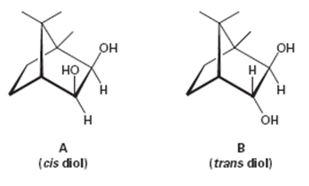
The observation that cis-diol A cleaved approximately I million times faster than the trans-diol B is to be explained.
Concept introduction:
The cleavage of
To explain:
The observation that cis-diol A cleaved approximately 1 million times faster than the trans-diol B is to be explained.
Trending nowThis is a popular solution!

Chapter 8 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- When warmed in dilute sulfuric acid, 1-phenyl-1,2-propanediol undergoes dehydration and rearrangement to give 2-phenylpropanal. (a) Propose a mechanism for this example of a pinacol rearrangement (Section 10.7). (b) Account for the fact that 2-phenylpropanal is formed rather than its constitutional isomer, 1-phenyl-1-propanone.arrow_forwardAn inexperienced graduate student treated dec-5-ene with borane in THF, placed the flask in a refrigerator, and left for aparty. When he returned from the party, he discovered that the refrigerator was broken and that it had gotten quite warminside. Although all the THF had evaporated from the flask, he treated the residue with basic hydrogen peroxide. Tohis surprise, he recovered a fair yield of decan-1-ol. Use a mechanism to show how this reaction might have occurred.(Hint: The addition of BH3 is reversible.)*8arrow_forward1) 1-bromobutane will undergo reactions when heated, as shown by reactions A and B CH3CH2CH2CH2Br A B CH3CH2CH2CH2OH CH3CH2CH=CH2 a)For reactions A and B give the reagents used in each case.b)Reaction A was repeated using 1-iodobutane instead of 1-bromobutane, Explain any difference in therate of reaction observed.C)What type of organic reaction is A?d) Show the mechanism for reaction Ae)Reaction A was repeated with 2-bromo-2-methylpropane instead of 1-bromobutane.i)Name the organic compound formedii) The mechanism of the reaction with 2-bromo-2-methylpropane differs from the mechanism ofreaction A. Describe how the mechanisms differ.f)What type of reaction is B?g)If reaction B was repeated with 2-bromobutane, name the other organic products that can form aswell…arrow_forward
- Propose a mechanism and use stability factors to account for the formation of a major product in the reaction of pyrrole with Br2, FeBr3.arrow_forwardComplete the reaction schemes below providing the reagents required to achieve thetransformation. More than one step may be necessary for each scheme.arrow_forward5. Optically pure 2-octyl sulfonate was treated with varying mixtures of water and dioxane, and the optical purity of the resulting product (2-octanol) was found to vary with the ratio of water to dioxane, as shown in the following table (J. Am. Chem. Soc. 1965, 87, 287-291). Given that dioxane possesses fairly nucleophilic oxygen atoms, provide a complete mechanism that explains the variation in in the product's optical purity due to changes in solvent composition. Hint, the solvent is doing more here than just dissolving.. it is partaking in the reaction. Solvent Ratio (water : dioxane) 25:75 50: 50 75: 25 100 :0 Optical purity of (S)-2-octanol 77% 88% 95% 100% H20 он (R)-2-Octyl sulfonate (optically pure) (S)-2-Octanol (dioxane)arrow_forward
- When bromomethane undergoes solvolysis in a solvent mixture composed of 90% water/10% acetone, the reaction rate is faster than when the same compound is solvolyzed in 80% water/20% acetone. Explain.arrow_forward12:53 ul 4G O 2. Show a reaction scheme with all the reactants and reagents of the nitration reaction of bromobenzene and show the major products(s), with valid reasons motivate why the positioning of the two constituents' favour ortho, para and/or meta positioning Add a caption... > Status (Custom) +arrow_forwardCompelling evidence for the existence of a tetrahedral intermediate in nucleophilic acyl substitution was obtained in a series of elegant experiments carried out by Myron Bender in 1951. The key experiment was the reaction of aqueous−OH with ethyl benzoate (C6H5COOCH2CH3) labeled at the carbonyl oxygen with 18O. Bender did not allow the hydrolysis to go to completion, and then examined the presence of a label in the recovered starting material. He found that some of the recovered ethyl benzoate no longer contained a label at the carbonyl oxygen. With reference to the accepted mechanism of nucleophllic acyl substitution, explain how this provides evidence for a tetrahedral intermediate.arrow_forward
- Trichloroisocyanuric acid, TCICA, also known as “swimming pool chlorine,” is a stable solid that oxidizes alcohols, following a mechanism similar to oxidation by HOCl. No reaction occurs between TCICA and the alcohol (in a solvent such as acetonitrile) until one drop of HCl(aq) is added, whereupon the reaction is over in a few minutes. Write the mechanism for this oxidation that shows the key role of the acid catalyst. Show the oxidation of just one alcohol, not three.arrow_forwardGive the major organic product(s) for each step of the following reactionsarrow_forward(i) OEt OEt 1) NaOEt 2) H3O* workup W 1) NaOEt 2) Br V Compound W can be synthesised in an excellent yield via an intramolecular Claisen reaction of 1,6-diester V. Draw the structure of product W and provide detailed reactions mechanisms to account for its formation. (ii) Draw the product X which would form upon the alkylation reaction of W with the reagents shown.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
