
a)
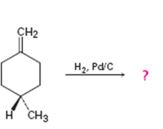
Interpretation:
Whether the reaction shown will result in an optically active product is to be stated.
Concept introduction:
The catalytic hydrogenation is a heterogeneous process that occurs with syn stereochemistry. Both the hydrogens add from the same face of the double bond from the catalytic surface.
To state:
Whether the reaction shown will result in an optically active product.
b)
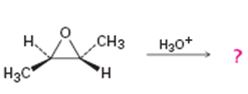
Interpretation:
Whether the reaction shown will result in an optically active product is to be stated.
Concept introduction:
The
To state:
Whether the reaction shown will result in an optically inactive product.
c)
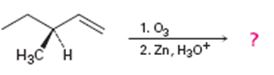
Interpretation:
Whether the reaction shown will result in an optically active product is to be stated.
Concept introduction:
During ozonolysis the double bond in
To state:
Whether the reaction shown will result in an optically active product.
Trending nowThis is a popular solution!

Chapter 8 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- What is the mechanism of the reaction below.arrow_forwardA carbamate can be prepared by treating an isocyanate with an alcohol, as shown here. This type of reaction is used to synthesize polyurethanes- polymers that have a wide variety of industrial applications, such as surface sealants, high-performance adhesives, and synthetic fibers. Propose a mechanism for this transformation. R'—ОН R. `N=C=0 R. OR' An isocyanate A carbamate (Substituted urethane) O=Carrow_forward. Predict the products of the following reactions. When more than one product is expected, predict which will be the major product?arrow_forward
- Which of the following is most likely to be the first step in the general mechanism for electrophilic aromatic substitution? H E E H (+) E + H H .E E + H + E + НЕarrow_forwardFor each of the reaction below, draw the curved arrows and lone pairs of electrons to show the mechanism. Predict which side of the reaction will be favoured under equilibrium conditions. Give a brief explanation of your prediction. NーHarrow_forwardSuggest a reasonable mechanism for the reaction shown Br here. HO CH;CH,OHarrow_forward
- 2) Draw a plausible mechanism for the following reaction by using the guidelines, the template, and the instructions below. N H H3O+ H H₂N H 0: H • 14 H H + H H Harrow_forwardWrite down the mechanism of the reactions given below and find the resulting product.arrow_forwardWhich of the following would likely to give racemic product upon substitution reaction with hydroxide ion and have [o]p=0? IV O O O Oarrow_forward
- Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?arrow_forwardWhich set of conditions for the reaction given below would provide the largest amounts of substitution products? Conditions? substitution products NACN in DME NAOH in Water O NACN in MeOH NaF in DMFarrow_forwardIf the above reaction was done with chlorine instead of bromine, how many productscould form (include stereoisomers)? Show all possible products.arrow_forward
