
Thermodynamics: An Engineering Approach
9th Edition
ISBN: 9781259822674
Author: Yunus A. Cengel Dr., Michael A. Boles
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 8.8, Problem 14P
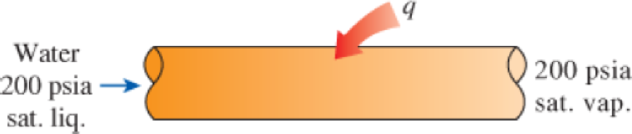
Saturated steam is generated in a boiler by converting a saturated liquid to a saturated vapor at 200 psia. This is done by transferring heat from the combustion gases, which are at 700°F, to the water in the boiler tubes. Calculate the wasted work potential associated with this heat transfer process. How does increasing the temperature of the combustion gases affect the work potential of the steam stream? Take T0 = 80°F and P0 = 14.7 psia.
FIGURE P8–14E
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The turbines in steam power plants operate essentially under adiabatic conditions. A plant engineer suggests ending this practice. She proposes to run cooling water through the outer surface of the casing to cool the steam as it flows through the turbine. This way, she reasons, the entropy of the steam will decrease, the performance of the turbine will improve, and as a result the work output of the turbine will increase. How would you evaluate this proposal?
A turbine generates 8000 kW at 40,000 kg/hr of steam and exhaust at 10 kPa with an enthalpy of 2510 kJ/kg. If the boiler pressure is 6 Mpa, determine the isentropic efficiency.
Refrigerant-134a at 1600 kPa and 80°C is expanded adiabatically in a closed system to 100 kPa with an isentropic expansion efficiency of 85 percent. Determine the second-law efficiency of this expansion. Take T0 = 25°C and P0 = 100 kPa.
Chapter 8 Solutions
Thermodynamics: An Engineering Approach
Ch. 8.8 - What final state will maximize the work output of...Ch. 8.8 - Is the exergy of a system different in different...Ch. 8.8 - Under what conditions does the reversible work...Ch. 8.8 - How does useful work differ from actual work? For...Ch. 8.8 - How does reversible work differ from useful work?Ch. 8.8 - Is a process during which no entropy is generated...Ch. 8.8 - Consider an environment of zero absolute pressure...Ch. 8.8 - It is well known that the actual work between the...Ch. 8.8 - Consider two geothermal wells whose energy...Ch. 8.8 - Consider two systems that are at the same pressure...
Ch. 8.8 - Prob. 11PCh. 8.8 - Does a power plant that has a higher thermal...Ch. 8.8 - Prob. 13PCh. 8.8 - Saturated steam is generated in a boiler by...Ch. 8.8 - One method of meeting the extra electric power...Ch. 8.8 - A heat engine that receives heat from a furnace at...Ch. 8.8 - Consider a thermal energy reservoir at 1500 K that...Ch. 8.8 - A heat engine receives heat from a source at 1100...Ch. 8.8 - A heat engine that rejects waste heat to a sink at...Ch. 8.8 - A geothermal power plant uses geothermal liquid...Ch. 8.8 - A house that is losing heat at a rate of 35,000...Ch. 8.8 - A freezer is maintained at 20F by removing heat...Ch. 8.8 - Prob. 24PCh. 8.8 - Prob. 25PCh. 8.8 - Prob. 26PCh. 8.8 - Can a system have a higher second-law efficiency...Ch. 8.8 - A mass of 8 kg of helium undergoes a process from...Ch. 8.8 - Which is a more valuable resource for work...Ch. 8.8 - Which has the capability to produce the most work...Ch. 8.8 - The radiator of a steam heating system has a...Ch. 8.8 - A well-insulated rigid tank contains 6 lbm of a...Ch. 8.8 - A pistoncylinder device contains 8 kg of...Ch. 8.8 - Prob. 35PCh. 8.8 - Prob. 36PCh. 8.8 - Prob. 37PCh. 8.8 - A pistoncylinder device initially contains 2 L of...Ch. 8.8 - A 0.8-m3 insulated rigid tank contains 1.54 kg of...Ch. 8.8 - An insulated pistoncylinder device initially...Ch. 8.8 - Prob. 41PCh. 8.8 - An insulated rigid tank is divided into two equal...Ch. 8.8 - A 50-kg iron block and a 20-kg copper block, both...Ch. 8.8 - Prob. 45PCh. 8.8 - Prob. 46PCh. 8.8 - Prob. 47PCh. 8.8 - A pistoncylinder device initially contains 1.4 kg...Ch. 8.8 - Prob. 49PCh. 8.8 - Prob. 50PCh. 8.8 - Prob. 51PCh. 8.8 - Air enters a nozzle steadily at 200 kPa and 65C...Ch. 8.8 - Prob. 54PCh. 8.8 - Prob. 55PCh. 8.8 - Argon gas enters an adiabatic compressor at 120...Ch. 8.8 - Prob. 57PCh. 8.8 - Prob. 58PCh. 8.8 - The adiabatic compressor of a refrigeration system...Ch. 8.8 - Refrigerant-134a at 140 kPa and 10C is compressed...Ch. 8.8 - Air enters a compressor at ambient conditions of...Ch. 8.8 - Combustion gases enter a gas turbine at 900C, 800...Ch. 8.8 - Steam enters a turbine at 9 MPa, 600C, and 60 m/s...Ch. 8.8 - Refrigerant-134a is condensed in a refrigeration...Ch. 8.8 - Prob. 66PCh. 8.8 - Refrigerant-22 absorbs heat from a cooled space at...Ch. 8.8 - Prob. 68PCh. 8.8 - Prob. 69PCh. 8.8 - Air enters a compressor at ambient conditions of...Ch. 8.8 - Hot combustion gases enter the nozzle of a...Ch. 8.8 - Prob. 72PCh. 8.8 - A 0.6-m3 rigid tank is filled with saturated...Ch. 8.8 - Prob. 74PCh. 8.8 - Prob. 75PCh. 8.8 - An insulated vertical pistoncylinder device...Ch. 8.8 - Liquid water at 200 kPa and 15C is heated in a...Ch. 8.8 - Prob. 78PCh. 8.8 - Prob. 79PCh. 8.8 - A well-insulated shell-and-tube heat exchanger is...Ch. 8.8 - Steam is to be condensed on the shell side of a...Ch. 8.8 - Prob. 82PCh. 8.8 - Prob. 83PCh. 8.8 - Prob. 84PCh. 8.8 - Prob. 85RPCh. 8.8 - Prob. 86RPCh. 8.8 - An aluminum pan has a flat bottom whose diameter...Ch. 8.8 - Prob. 88RPCh. 8.8 - Prob. 89RPCh. 8.8 - A well-insulated, thin-walled, counterflow heat...Ch. 8.8 - Prob. 92RPCh. 8.8 - Prob. 93RPCh. 8.8 - Prob. 94RPCh. 8.8 - Prob. 95RPCh. 8.8 - Nitrogen gas enters a diffuser at 100 kPa and 110C...Ch. 8.8 - Prob. 97RPCh. 8.8 - Steam enters an adiabatic nozzle at 3.5 MPa and...Ch. 8.8 - Prob. 99RPCh. 8.8 - A pistoncylinder device initially contains 8 ft3...Ch. 8.8 - An adiabatic turbine operates with air entering at...Ch. 8.8 - Steam at 7 MPa and 400C enters a two-stage...Ch. 8.8 - Prob. 103RPCh. 8.8 - Steam enters a two-stage adiabatic turbine at 8...Ch. 8.8 - Prob. 105RPCh. 8.8 - Prob. 106RPCh. 8.8 - Prob. 107RPCh. 8.8 - Prob. 108RPCh. 8.8 - Prob. 109RPCh. 8.8 - Prob. 111RPCh. 8.8 - A passive solar house that was losing heat to the...Ch. 8.8 - Prob. 113RPCh. 8.8 - A 4-L pressure cooker has an operating pressure of...Ch. 8.8 - Repeat Prob. 8114 if heat were supplied to the...Ch. 8.8 - Prob. 116RPCh. 8.8 - A rigid 50-L nitrogen cylinder is equipped with a...Ch. 8.8 - Prob. 118RPCh. 8.8 - Prob. 119RPCh. 8.8 - Prob. 120RPCh. 8.8 - Reconsider Prob. 8-120. The air stored in the tank...Ch. 8.8 - Prob. 122RPCh. 8.8 - Prob. 123RPCh. 8.8 - Prob. 124RPCh. 8.8 - Prob. 125RPCh. 8.8 - Prob. 126RPCh. 8.8 - Prob. 127RPCh. 8.8 - Water enters a pump at 100 kPa and 30C at a rate...Ch. 8.8 - Prob. 129RPCh. 8.8 - Prob. 130RPCh. 8.8 - Obtain a relation for the second-law efficiency of...Ch. 8.8 - Writing the first- and second-law relations and...Ch. 8.8 - Prob. 133RPCh. 8.8 - Keeping the limitations imposed by the second law...Ch. 8.8 - Prob. 135FEPCh. 8.8 - Prob. 136FEPCh. 8.8 - Prob. 137FEPCh. 8.8 - Prob. 138FEPCh. 8.8 - A furnace can supply heat steadily at 1300 K at a...Ch. 8.8 - A heat engine receives heat from a source at 1500...Ch. 8.8 - Air is throttled from 50C and 800 kPa to a...Ch. 8.8 - Prob. 142FEPCh. 8.8 - A 12-kg solid whose specific heat is 2.8 kJ/kgC is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Similar questions
- An isentropic (s1 = s2) turbine in a steam generating plant receives steam at a pressure of 7 MPa and 550 0C, and exhaust at 20 kPa. The turbine inlet is 3 m higher than the turbine exit. The inlet steam velocity is 15 m/sec and the exit velocity is 300 m/sec. Calculate the turbine work per unit mass of steam. (Hint: s1 = s2 = sf2 + x2sfg2)arrow_forwardRefrigerant-134a is to be cooled by water in a condenser. The refrigerant enters the condenser with a mass flow rate of 6 kg/min at 1 MPa and 70°C and leaves at 35°C. The cooling water enters at 300 kPa and 15°C and leaves at 25°C. Neglecting any pressure drops, determine the mass flow rate of the cooling water required.arrow_forwardSteam at 6 MPa and 500°C enters a two-stage adiabatic turbine at a rate of 15 kg/s. Ten percent of the steam is extracted at the end of the first stage at a pressure of 1.2 MPa for other use. The remainder of the steam is further expanded in the second stage and leaves the turbine at 20 kPa. Determine the power output of the turbine, assuming the turbine has an isentropic efficiency of 88 percent.arrow_forward
- Determine the isentropic efficiency of the rotary engine and the second law efficiency of therotary engine when the Water vapor inlet a rotary engine at 500 oC temperature and 1200 kPa pressure and exits at 300 oC temperature and 200 kPa pressure.. According to this: (take close temperature of 25 0C)arrow_forwardIn large steam power plants, the feedwater is frequently heated in a closed feedwater heater by using steam extracted from the turbine at some stage. Steam enters the feedwater heater at 1 MPa and 200°C and leaves as saturated liquid at the same pressure. Feedwater enters the heater at 2.5 MPa and 50°C and leaves at 10°C below the exit temperature of the steam. Determine the ratio of the mass flow rates of the extracted steam and the feedwater.arrow_forwardSteam enters an adiabatic turbine at 2.5MPa pressure and 450C, and at 60kPa pressure and 100C. comes out. If the power output of the turbine is 3MW, the mass flow and the isentropic efficiency of the turbine calculate.arrow_forward
- A plant engineer suggests ending this practice. She proposes to run cooling water through the outer surface of the casing to cool the steam as it flows through the turbine. This way, she reasons, the entropy of the steam will decrease, the performance of the turbine will improve, and as a result the work output of the turbine will increase. How would you evaluate this proposal?arrow_forwardAir is expanded in an adiabatic turbine of 85 percent isentropic efficiency from an inlet state of 2200 kPa and 300C to an outlet pressure of 200 kPa. Calculate the outlet temperature of air and the work produced by this turbine per unit mass of air.arrow_forwardAn adiabatic air compressor is to be powered by a direct-coupling adiabatic steam turbine that is also driving a generator. Steam enters the turbine at 12.5 MPa and 900 oC at a rate of 27 kg/s and exits at 4 kPa and a quality of 0.6. Air enters the compressor at 98 kPa and 310 K at a rate of 7 kg/s and exits at 1 MPa and 700 K. Determine the net power delivered to the generator by the turbine. h1 (kJ/kg) h2 (kJ/kg) Wcomp,in (kW) Wturb,in (kW) Wnet (kW)arrow_forward
- Steam at 100 psia and 650F is expanded adiabatically in a closed system to 10 psia. Determine the work produced, in Btu/lbm, and the final temperature of steam for an isentropic expansion efficiency of 80 percent.arrow_forwardWhat is the minimum internal energy that steam can achieve as it is expanded adiabatically in a closed system from 1500 kPa and 320°C to 100 kPa?arrow_forwardConsider an adiabatic turbine operating steadily. Does the work output of the turbine have to be equal to the decrease in the energy of the steam flowing through it?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY

Elements Of Electromagnetics
Mechanical Engineering
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:9780134319650
Author:Russell C. Hibbeler
Publisher:PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:9781259822674
Author:Yunus A. Cengel Dr., Michael A. Boles
Publisher:McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:9781118170519
Author:Norman S. Nise
Publisher:WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:9781337093347
Author:Barry J. Goodno, James M. Gere
Publisher:Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:9781118807330
Author:James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:WILEY
Extent of Reaction; Author: LearnChemE;https://www.youtube.com/watch?v=__stMf3OLP4;License: Standard Youtube License