
Concept explainers
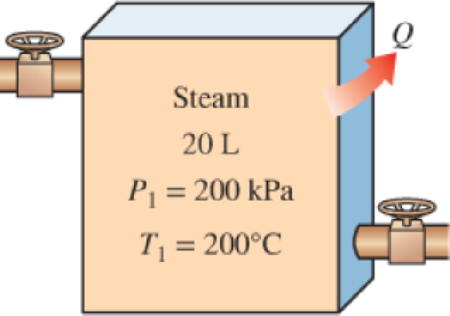
The radiator of a steam heating system has a volume of 20 L and is filled with superheated water vapor at 200 kPa and 200°C. At this moment both the inlet and the exit valves to the radiator are closed. After a while it is observed that the temperature of the steam drops to 80°C as a result of heat transfer to the room air, which is at 21°C. Assuming the surroundings to be at 0°C, determine (a) the amount of heat transfer to the room and (b) the maximum amount of heat that can be supplied to the room if this heat from the radiator is supplied to a heat engine that is driving a heat pump. Assume the heat engine operates between the radiator and the surroundings.
FIGURE P8–31
Trending nowThis is a popular solution!

Chapter 8 Solutions
Thermodynamics: An Engineering Approach
- What is the specific internal energy of water at 50 kPa and 200°C?arrow_forward2 kg of air at 298 K and 120 kPa inside a piston cylinder device is heated to 860 K in an isochoric process and then, it is heated to 1200 K in an isobaric manner. Determine the overall heat transfer during the process? 1636 kJ 1436 kJ 1200 kJ 1000 kJ nonearrow_forwardWhat is an adiabatic process? What is an adiabatic system?arrow_forward
- A 0.8-m3 rigid tank contains carbon dioxide (CO2) gas at 250 K and 100 kPa. A 500-W electric resistance heater placed in the tank is now turned on and kept on for 40 min, after which the pressure of CO2 is measured to be 175 kPa. Assuming the surroundings to be at 300 K and using constant specific heats, determine the final temperature of CO2.arrow_forwardA rigid vessel contains 5.0 kg of wet steam at 0.4 MPa. After the addition of 9585 kJ the steam has a pressure of 2.0 MPa and a temperature of 700°C. Determine the initial internal energy and the specific volume of the steam.arrow_forwardThe radiator of a steam heating system has a volume of 20 L and is filled with superheated water vapor at 200 kPa and 200°C. At this moment both the inlet and the exit valves to the radiator are closed. After a while it is observed that the temperature of the steam drops to 80°C as a result of heat transfer to the room air, which is at 21°C. Assuming the surroundings to be at 0°C, determine the amount of heat transfer to the room.arrow_forward
- The volume of 1 kg of helium in a piston–cylinder device is initially 5 m3 . Now helium is compressed to 2 m3 while its pressure is maintained constant at 130 kPa. Determine the initial and final temperatures of helium as well as the work required to compress it, in kJ.arrow_forwardA 0.8-m3 rigid tank contains carbon dioxide (CO2) gas at 250 K and 100 kPa. A 500-W electric resistance heater placed in the tank is now turned on and kept on for 40 min, after which the pressure of CO2 is measured to be 175 kPa. Assuming the surroundings to be at 300 K and using constant specific heats, determine the net amount of heat transfer from the tank.arrow_forwardA 2-ft3 rigid tank contains saturated refrigerant- 134a at 160 psia. Initially, 5 percent of the volume is occupied by liquid and the rest by vapor. A valve at the top of the tank is now opened, and vapor is allowed to escape slowly from the tank. Heat is transferred to the refrigerant such that the pressure inside the tank remains constant. The valve is closed when the last drop of liquid in the tank is vaporized. Determine the total heat transfer for this process.arrow_forward
- Consider two identical rooms, one with a 2-kW electric resistance heater and the other with three couples fast dancing. In which room will the air temperature rise more quickly?arrow_forwardAn inventor claims to have devised a process using only saturated steam at 100C which by a complicated series of steps, make heat continuously available at T of 177C. He claims further that, for every kilogram of steam taken into the process, 1800 kJ of energy as heat is liberated at this higher T of 177C. Show whether or not such a process is possible. In order to give the inventor the benefit of any doubt, assume cooling water to be available in unlimited quantity at 0C. topic: introduction to chemical engineerint thermodynamicsarrow_forwardNitrogen at 100 psia and 300°F in a rigid container is cooled until its pressure is 50 psia. Determine the work done and the heat transferred during this process, in Btu/lbmarrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





