
a)
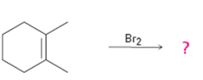
Interpretation:
The product formed with its stereochemistry, when 1,2-dimethylcyclohexene reacts with Br2 is to be given. The mechanism of its formation also is to be given.
Concept introduction:
The reaction of halogens to
To give:
The product expected, along with its stereochemistry, when Br2 is added to 1,2-dimethylcyclohexene.
b)
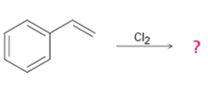
Interpretation:
The product obtained with its stereochemistry, in the reaction shown, is to be given. The mechanism of its formation also is to be given.
Concept introduction:
The reaction of halogens to alkenes occurs with anti stereochemistry, that is, the two halogen atoms come from opposite faces of the double bond, one from top face and other from bottom face. In the first step, the addition of halogen to the double bond in the alkenes results in the formation of a cyclic halonium ion with the simultaneous elimination of a halide ion. The large halonium ion shields one side of the molecule. Hence the attack of the halide ion occurs from the opposite, unshielded side to yield a trans product.
To give:
The product expected, along with its stereochemistry, when Cl2 is added to styrene (vinyl benzene).
c)
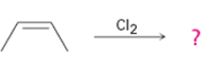
Interpretation:
The product obtained with its stereochemistry in the reaction shown is to be given. The mechanism of its formation also is to be given.
Concept introduction:
The reaction of halogens to alkenes occurs with anti stereochemistry, that is, the two halogen atoms come from opposite faces of the double bond- one from top face and other from bottom face. In the first step, the addition of halogen to the double bond in the alkenes results in the formation of a cyclic halonium ion with the simultaneous elimination of a halide ion. The large halonium ion shields one side of the molecule. Hence the attack of the halide ion occurs from the opposite, unshielded side to yield a trans-product.
To give:
The product expected, along with its stereochemistry, when Cl2 is added to 2-butene.
Trending nowThis is a popular solution!

Chapter 8 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
- Predict the product(s) and provide the mechanism for each reaction below.arrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardBenzyl bromide is converted into benzaldehyde by heating in dimethyl sulfoxide. Propose a structure for the intermediate, and show the mechanisms of the two steps in the reaction.arrow_forward
