
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 18PP
PRACTICE PROBLEM 9.18
What are the expected ratios of the
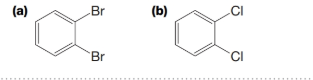
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A colorless solid that melts at 103 to 105 C was subjected to IR Spectroscopy to determine the functional groups present and partially determine its nomenclature based on the molecular formula given.
What are the functional groups present based on the major infrared absorptions? (Select Possible Answers)
Name the following compounds A and B.
How could you distinguish these two molecules by using 1H NMR and IR techniques?
Propose an analytical technique to determine the iron content of these compounds.
Calculate the mass percentages of C and H of compound B (C: 12.01 g/mol; H: 1.008 g/mol; Fe: 55.845 g/mol).
What is the possible unknown compound based on the IR Spectrum, please give detailed explanation on why it is the compound and also give detailed explanation on each of the other compounds
unknown compound melting point is 170 0C, also if you have answered it before please do not answer again, it is not for you
Chapter 9 Solutions
Organic Chemistry
Ch. 9 - Prob. 1PPCh. 9 - PRACTICE PROBLEM 9.2 What compound with molecular...Ch. 9 - PRACTICE PROBLEM 9.3
Using the method of Section...Ch. 9 - PRACTICE PROBLEM 9.4 How many signals would each...Ch. 9 - Prob. 5PPCh. 9 - Prob. 6PPCh. 9 - PRACTICE PROBLEM 9.7
The relative chemical shifts...Ch. 9 - Prob. 8PPCh. 9 - PRACTICE PROBLEM 9.9 Propose a structure for...Ch. 9 - PRACTICE PROBLEM 9.10
What is the dihedral angle...
Ch. 9 - PRACTICE PROBLEM 9.11 Draw the most stable chair...Ch. 9 - Prob. 12PPCh. 9 - PRACTICE PROBLEM 9.13 How many signals would you...Ch. 9 - Prob. 14PPCh. 9 - Prob. 15PPCh. 9 - Prob. 16PPCh. 9 - Prob. 17PPCh. 9 - PRACTICE PROBLEM 9.18
What are the expected ratios...Ch. 9 - Prob. 19PPCh. 9 - How many 1H NMR signals (not peaks) would you...Ch. 9 - How many 13C NMR signals would you predict for...Ch. 9 - Prob. 22PCh. 9 - Prob. 23PCh. 9 - Prob. 24PCh. 9 - Compound Q has the molecular formula C7H8. The...Ch. 9 - 9.26 Explain in detail how you would distinguish...Ch. 9 - Compound S (C8H16) reacts with one mole of bromine...Ch. 9 - A compound with molecular formula C4H8O has a...Ch. 9 - In the mass spectrum of 2, 6-dimethyl-4-heptanol...Ch. 9 - Prob. 30PCh. 9 - What are the masses and structures of the ions...Ch. 9 - Prob. 32PCh. 9 - Ethyl bromide and methoxybenzene (shown below)...Ch. 9 - 9.34 The homologous series of primary amines, ,...Ch. 9 - Propose a structure that is consistent with each...Ch. 9 - 9.36 Propose structures for compounds E and F....Ch. 9 - 9.37 Use the NMR and IR data below to propose a...Ch. 9 - 9.38 When dissolved in , a compound (K) with the...Ch. 9 - Compound T (C5H8O) has a strong IR absorption band...Ch. 9 - Prob. 40PCh. 9 - Deduce the structure of the compound that gives...Ch. 9 - Deduce the structure of the compound that gives...Ch. 9 - The 1H NMR spectrum of a solution of 1,...Ch. 9 - Acetic acid has a mass spectrum showing a...Ch. 9 - The 1H NMR peak for the hydroxyl proton of...Ch. 9 - The 1H NMR study of DMF (N, N-dimethylformamide)...Ch. 9 - 9.48 The mass spectra of many benzene derivatives...Ch. 9 - Prob. 49PCh. 9 - 1. Given the following information, elucidate the...Ch. 9 - Two compounds with the molecular formula C5H10O...
Additional Science Textbook Solutions
Find more solutions based on key concepts
For the 3pz and 4dxz hydrogen-like atomic orbitals, sketch the following: a. The radial function R b. The radia...
Inorganic Chemistry
If a compound has a molecular ion with an odd-numbered mass, then the compound contains an odd number of nitrog...
Organic Chemistry
The glycine cleavage system is a group of four enzymes that together catalyze the following reaction: glycine+T...
Organic Chemistry (8th Edition)
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
The NF bond is more polar than the NH bond: but NF3 has a smaller dipole moment than NH3. Explain this curious ...
Organic Chemistry (9th Edition)
Strengths of Brnsted-Lowry Acids and Bases Consider the following: CO/ is a weaker base than hydroxide ion, and...
Chemistry: The Molecular Nature of Matter
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the structure of the compound that produced the spectra below. The infrared spectrum has strong bands at 1720 and 1738 cm-1.arrow_forwardIm having a hard time distinguishing peaks on an IR spectrum. Theyre often very crowded, and i dont know if broad peaks get combined with sharp peaks. On this IR spectrum, it seems there is a broad range, medium strength band extending from 4000 to (about) 2000 cm-1, with several sharp peaks on top of that range. Does this mean that there is a carboxylic acid O-H present as well as the other bonds determined by the sharp peaks?arrow_forward2) Figures 2 to 4 show the IR, 1H NMR and 13C NMR spectra of a compound with formula C3H5N 2a) Identify bands A and B in the IR spectrum 2b) Using the information from the 1H NMR spectrum, fill in the table below Peak cluster (ppm) Multiplicity Number of H in neighbouring C Proposed identification 0 1.30 2.35 2c) Propose a structure for the compound C3H5N. Justify your answer by referring to key spectral features of the 3 spectra.arrow_forward
- Please discuss the following IR spectrum of the ferrocene compound after it was recrystallisedarrow_forwardThe cyclobutenyl dichloride below reacts with the powerful Lewis acid antimony pentafluoride in liquid SO2 at -75o to give a pale yellow solution that exhibits one singlet at 3.68 ppm in its 1H-NMR spectrum. The species in solution has been deduced to be the salt C8H12X2 where X is an hexahaloantimonate anion. Draw the cation part of the salt C8H12X2arrow_forwardExplain why a basic solution of methyl ethylamine is neutralized by the addition of a concentrated hydrochloric acid, its weak absorption band centered at 244 nm starts to disappeararrow_forward
- The IR spectrum, 1H NMR spectrum, and 13C NMR spectrum for the unknown compound with the formula C5H12O are given below. Make a structure for the unknown and assign the peaks in the spectra by the provided tables.arrow_forwardA proton nmr spectrum is shown for a compound having formula C5H9NO4 the infrared spectrum shows strong band at 1750 andarrow_forwardInterpet and analyze this Infrared (IR) spectra to show the signs from the peaks and what they tell about the unknown compound.arrow_forward
- 1.) Provide the correct answers in the given statement with the given compounds in the image. A. In separate test tubes, tert-butyl chloride in AlCl3 was added. Which compound/s will yield a change in the color of the AlCl3 crystal? B. Which compound/s will react with Br2 in water under light conditions? C. Which compound/s will react with I2 in KI under dark conditions?arrow_forwardSuggest structures given the 1H NMR spectra and formulas for each of the compounds below. C9H10Oarrow_forwardSuggest structures given the 1H NMR spectra and formulas for each of the compounds below. C10H14, two peak area ratio is 1:6arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY