
(a)
Interpretation:
The
Concept Introduction:
Polymers are macromolecules which are formed a by number of repeating units called monomers.
(b)
Interpretation:
The reasons why repeating head-to-tail arrangement is not possible in Teflon has to be identified.
Concept Introduction:
Polymers: Monomers combine together to form polymers. Monomers are the repeating units of small molecules which link together to form polymers and the process is called as polymerization.
Polymers are macromolecules which are formed a by number of repeating units called monomers.
Terpenes are made by joining five-carbon units, usually in a head to tail-fashion.
Isoprene unit:
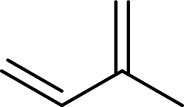
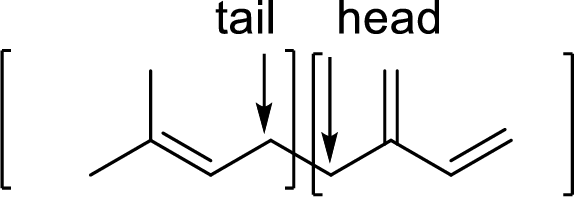
Branched end of isoprene – Head
Unbranched end of isoprene - Tail
(c)
Interpretation:
The characteristics that Teflon and
Concept Introduction:
Polymers are macromolecules which are formed a by number of repeating units called monomers.
Polymers: Monomers combine together to form polymers. Monomers are the repeating units of small molecules which link together to form polymers and the process is called as polymerization.
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Chemistry In Context
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning





