
Concept explainers
If lactic acid is similar in strength to acetic acid (Table. 9-2), predict whether reactants or products are favored in each reaction. 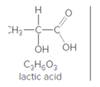
a.
lactic acid
b.
lactic acid
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
General, Organic, and Biological Chemistry - 4th edition
- What is the ionization constant at 25 C for the weak acid CH3 NH3+, the conjugate acid of the weak base CH3NH2, Kb=4.4104.arrow_forwardwhat is the value of k for the reverse reaction if K forward + 6.49 x 10^-2arrow_forwardAmmonia, NH3, is a base and will readily accept a proton in accordance with the following reaction: NH3(aq) + H2O <-> NH4+ + OH- Calculate the equilibrium constant for this reaction (the reaction goes from left to right) at 25 ºC. ∆G0NH3(ag) = -26.6 kJ/mol ∆G0H2O = -237.1 kJ/mol ∆G0NH4+ = -79.3 kJ/mol ∆G0OH- = -157.2 kJ/molarrow_forward
- What is the [H+] for a 0.010 M HOBr, hypobromous acid, solution? Ka for HOBr is 2.5 x 10-9. a) 2.5 x10-11 M b) 2.5 x10-7 M c) 5.0 x 10-7 M d) 5.0 x 10-6 M e) 5.0 x 10-5 Marrow_forwardThe Ka of acetic acid is 1.80 x 10-5. A. What is the pH of a 0.0010 M solution of acetic acid? B. What are the equilibrium concentrations of the weak acid and its conjugate base?arrow_forwardWhat is the pH of a balsamic vinegar having an acetic acid (M.W. = 60.05g/mol; Ka = 1.8 x 10^-5) content of 5% (w/v)? w/v = weight of acetic acid/volume of vinegar.arrow_forward
- Nicole separately prepared solutions of FeSO4 and FeBr3 with concentrations 7.71×10-1 M and 3.77×10-2 M respectively. Calculate the p-value for all ions and substances in these solutions.arrow_forwardA 0.11 M solution of weak acid, HA, has a pH of 2.07.a. Write the dissociation reaction of the acid in water.b. Write the acid dissociation constant expression.c. Given the table below, what is the most probable identity of the acid?Acid KaHypochlorous acid 3.0 x 10⁻⁸Nitrous acid 7.1 x 10⁻⁴Hydrogen cyanide 6.2 x 10⁻10Propionic acid 1.3 x 10⁻⁵arrow_forwardBoric acid, BOH is used as a mild antiseptic. What is the PH of a 0.025 aqueous solution of boric acid? What is the degree of ionisation of boric acid in this solution? The hydrogen ion arises principally from the reaction: B(OH)3(aq) + H2O(l) > B(OH)4- (aq) + H+ (aq) The equilibrium constant for the reaction is 5.9 x10-10arrow_forward
- Boric acid, BOH is used as a mild antiseptic. What is the pH of a 0.025 aqueous solutionof boric acid? What is the degree of ionisation of boric acid in this solution? Thehydrogen ion arises principally from the reaction:B(OH)3(aq) + H2O(l) B(OH)4(aq)_+ H+(aq)The equilibrium constant for this reaction is 5.9x10-10arrow_forwardFor the reaction shown below, predict whether the reactants or the products are predominant at equilibrium. HCN (aq) + H2O (l) ⇄ CN- (aq) + H3O+ (aq) Keq = 4.9 x 10^-10 M Can you help me, please? Can you explain to me?arrow_forwardThe pigment cyanidin aglycone is one of the anthocyanin molecules that gives red cabbage (Brassica oleracea var. capitata f. rubra) its characteristic red coloration. Many chemistry students have used this red cabbage indicator to study acid-base chemistry. Estimate tire pH range at which cyanidin agly-cone shows a color change. Anth-H(aq) Anth(aq) + H+ (aq) Ka = 1.3 107arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





