
(a)
To Write: The formula for the total zero-point energy of all the modes of the field inside the box in terms of a triple integral over the mode numbers in the x, y, and z direction.
(a)
Answer to Problem 24P
Explanation of Solution
Formula used:
Total energy within the box can be written as:
Where,
Factor of
Calculation:
Wavelength can be given as:
So, speed of light:
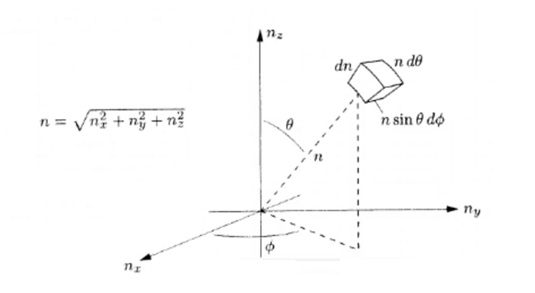
So,
From equation (1)
Conclusion:
The formula for the total zero-point energy of all the modes of the field inside the box is
(b)
To Show: The Planck length indeed has units of length.
To Calculate:Numerical value of Planck length.
(b)
Answer to Problem 24P
Explanation of Solution
Given:
Planck Length =
Formula used:
Dimension formula of
Calculation:
Dimension formula for given expression can be calculated as:
Planck length can be calculated as:
Conclusion:
It is proved that the Planck length indeed has units of length and its magnitude is
(c)
To Estimate: The energy per unit volume in empty state and equivalent mass density.
(c)
Answer to Problem 24P
Explanation of Solution
Formula used:
Where,
Calculation:
But,
Therefore,
Now, mass density can be calculated as
It is much greater than the average mass density of ordinary matter in the universe.
Conclusion:
Average mass density is
Want to see more full solutions like this?
Chapter A Solutions
An Introduction to Thermal Physics
- A function of the form e^−gx2 is a solution of the Schrodinger equation for the harmonic oscillator, provided that g is chosen correctly. In this problem you will find the correct form of g. (a) Start by substituting Ψ = e^−gx2 into the left-hand side of the Schrodinger equation for the harmonic oscillator and evaluating the second derivative. (b) You will find that in general the resulting expression is not of the form constant × Ψ, implying that Ψ is not a solution to the equation. However, by choosing the value of g such that the terms in x^2 cancel one another, a solution is obtained. Find the required form of g and hence the corresponding energy. (c) Confirm that the function so obtained is indeed the ground state of the harmonic oscillator and has the correct energy.arrow_forwardSketch the potential energy function of an electron in a hydrogen atom, (a) What is the value of this function at r=0 ? in the limit that r=? (b) What is unreasonable or inconsistent with the former result?arrow_forwardSchrodinger’s equation is an example of an eigenvalue equation. Which of the following functions is an eigenfunction of the operator x*d/dx?a. e-3xb. sin(3x2)c. ln(3x2)d. 3x2e. None of the abovearrow_forward
- Schrodinger’s equation is an example of an eigenvalue equation. Which of the following functions is an eigenfunction of the operator (1/x)d/dx?a. e-3xb. sin(3x2)c. ln(3x2)d. 3x2e. None of the abovearrow_forwardThe wavefunction for a quantum particle tunnelling through a potential barrier of thickness L has the form ψ(x) = Ae−Cx in the classically forbidden region where A is a constant and C is given by C^2 = 2m(U − E) /h_bar^2 . (a) Show that this wavefunction is a solution to Schrodinger’s Equation. (b) Why is the probability of tunneling through the barrier proportional to e ^−2CL?arrow_forwardQ/ Show that the oscillating probability density is linear in arbitrary wave beams that oscillates with a frequency equal to that of a linear oscillator?arrow_forward
- Consider an object containing 6 one-dimensional oscillators (this object could represent a model of 2 atoms in an Einstein solid). There are 4 quanta of vibrational energy in the object. (a) How many microstates are there, all with the same energy? (b) If you examined a collection of 38000 objects of this kind, each containing 4 quanta of energy, about how many of these objects would you expect to find in the microstate 000004?arrow_forward1. a. For a free particle, write the relations between the wave vector k and itsmomentum vector p and angular frequency ω and its energy E.b. What is the general form in one dimension of the wave function for a freeparticle of mass m and momentum p?c. Can this wave function ever be entirely real? If so, show how this ispossible. If not, explain why not.d. What can you say about the integral of the |Ψ (x; t)|^2 from - ∞ to + ∞ ?e. Is this a possible wave function for a real, physical particle? Explain whyor why not.arrow_forwardDerive the Nernst Equation from the definition of the free energy, G.arrow_forward
- The wavefunction of is Ψ(x) = Axe(−ax^2)/2 for with energy E = 3aℏ2/2m. Find the bounding potential V(x). Looking at the potential’s form, can you write down the two energy levels that are immediately above E?arrow_forwardThe radial wave function of a quantum state of Hydrogen is given by R(r)= (1/[4(2π)^{1/2}])a^{-3/2}( 2 - r/a ) exp(-r/2a), where a is the Bohr radius.(a) Determine the radial probability density P(r) associated with the quantum state in question. (b) Show that the function P(r) you determined in part (a) is properly normalized.arrow_forwardThe condition of the rigid boundaries demands that the wave function should vanish for x=0 and for x=L because?arrow_forward
 Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning
Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax




