
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter F, Problem 2PP
Interpretation Introduction
Interpretation:
The products of the given reactions are to be given.
Concept introduction:
Thiols and disulphides undergo
This can be depicted as:
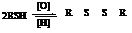
Sulphur ylide acts as a nucleophile and attacks the carbonyl carbon of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
OChem Question:
Starting with propyne and using any other required reagents, show how you would synthesize
(2R, 3R)- and (2S, 3S)-2,3-dibromobutane (as a racemic form)
Practice problem 1: Show the correct major organic products. Thank you !
What advantages and disadvantages of N-formylation? Why do we make it?
please
1. abstract
2. introduction
3. methodology
4.result and discuss
5. conclusion
Chapter F Solutions
Organic Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is major product choose from A, B, C, D , Earrow_forwardWhich of the following diols ARE likely to undergo oxidative change with H2CrO4? Select all that applyarrow_forwardWhat reagents are required to carry out the following reaction (select all that apply)? a HNO3 b H2SO4 c Cl2 d Br2 e H2O f CH3COCl g AlCl3 h FeCl3 i FeBr3 j CH3CHClCH3arrow_forward
- Problem B: Dicholorodiflouromethane, CCl2F2, a known refrigerant, can be produced by the chemical equation shown below. Consider the following: atomic mass of C is 12, Cl is 35, and F is 19. CH4 + Cl2 -> CCl4 + HCl (not balanced) CCl4 + HF -> CCl2F2 + HCl (not balanced) 1. Assuming that all the CCl4 produced in the first reaction is consumed in the second, how many moles of Cl2 must be consumed in the first reaction to produce 2.25kg CCl2F2 in the second? Show the complete solution A. 56.24 mol Cl2 B. 56.25 mol Cl2 C. 56.26 mol Cl2 D. 56.27 mol Cl2 E. 2250g Cl2 F. None of the abovearrow_forwardHello, I do not understand these questions and I am stuck. May I get help please?? Question: draw the MAJOR organic product(s) for the following reactions. If no reaction occurs state no reaction in the box providedarrow_forwardOrganic Chemistry question; Please provide a well explained and correct answer for the following. In Williamson ether synthesis reaction, NaH plays both roles as base and deprotonation to alcohol. How about other chemicals, and can they play the same role ? For instance, 1. Regular strong base, e.g. NaOH, KOH 2. Salt of strong base, weak acid, e.g. CH3COONa 3. Halide acid, e.g. HCl, HBr, HI 4. Salt of alcohol, e.g. CH3ONaarrow_forward
- true or false question. ( explanation not needed) a) Gilman copper reagent does 1,2 nucleophilic addition to α,β.unsaturated ketones b)Tertiary alcohol will be formed by the reaction between any ketone and Grignard. c)Cyclopentadiene anion is aromatic d)Tertiary alcohols undergoes dehydration by E1 mechanism e)1,4-addition product is the kinetic product in the addition of conjugated diene f) Benzene is more reactive than toluene in aromatic electrophilic substitution reactionarrow_forwardHello, May get help with this organic chemistry please? Question: Synthesize the following aromatic from benzenearrow_forwardIdentify the products that are expected in the given reaction. (select all that apply)arrow_forward
- Ochem prelab question I"ve attached part of the lab, but basically we are reducing m-nitroacetophenone to m-aminoacetophenone using granular tin. I've also attached one of my workups for the prelab, which require us to show the half reaction equation for reduction, the half reaction equation for oxidation and the overall balanced equation. Then determine the limiting reactant and theoretical yield Can you please check my work... especially my half reactions and see if those are correct (I'm not sure about the added e- and H+ Also, is the limiting reactant tin or should it be m-nitroacetophenonearrow_forwardidentify the reagents a-b in the following scheme: Please provide only typed answer solution no handwritten solution needed allowedarrow_forwardHello, I do not understand these questions and I am stuck. May I get help please please?? Question: Synthesize the following compounds using the rules provided using the alkyne shown and other compounds each containing 1 carbon must be incorporated into structure.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY