
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter F, Problem 3PP
Interpretation Introduction
Interpretation:
The given allyl disulfide is to be synthesised from allyl bromide.
Concept introduction:
Thiols and disulphides undergo
This can be depicted as:
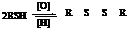
Sulphur ylide acts as a nucleophile and attacks the carbonyl carbon of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Problem 2: Draw the structures corresponding to the following names
2-bromo-5-chloro-3-methylhexane
10. Question shown in photo
Vanillin (Figure 5, C8H8O3) is an organic compound extracted from the vanilla bean and is used to add flavor to drinks, food, and pharmaceuticals. While natural vanilla extract is composed of many different compounds, artificial vanillin is usually synthesized as a pure compound. As of 2001 only about 15% of the annual demand for vanillin was isolated as a natural product. Identify the hybridization of the labeled atoms in vanillin.
a) i (sp2 ); ii (sp2 ); iii (sp2 )
b) i (sp2 ); ii (sp2 ); iii (sp3 )
c) i (sp2 ); ii (sp3 ); iii (sp2 )
d) i (sp3 ); ii (sp2 ); iii (sp2 )
How many straight-chain isomers can you write that fit the following descriptions? See Worked Example 12.12 for guidance.(a) Alcohols (-OH) with a longest chain of 6 carbons(b) Amines (-NH2)with a longest chain of 7 carbons
Chapter F Solutions
Organic Chemistry
Knowledge Booster
Similar questions
- All part provide compound name plz thank you plz IUPAC and normalarrow_forwardProvide a detailed step-by-step mechanism for the reaction below. Show each intermediate species. Use arrows to show the flow of electrons and clearly indicate any atoms with formal charges. I need help with this practice problem, thank you!arrow_forwardCH16 PE4 Please help Organic Chemistry problemarrow_forward
- Draw a skeletal structure of the following carbocation. Identify it as primary, secondary, or tertiary, and identify the hydrogen atoms that have the proper orientation for hyperconjugation in the conformation shown.arrow_forwardPractice problem 1: Show a full octet resonance contributor for thymine, using curved arrows to show its contributor. Add all formal charges, if necessary.arrow_forwardWhat is the name of the following alkyanes? Please show work/ explaination. Thank you!arrow_forward
- Which of the following are molecular formulas? Select all that apply. Group of answer choices C3H6O3 C2H6O C2H4O2 C3H9O2 CH2Oarrow_forwardProblem (#2.) For each ion below, draw all reasonable resonance structures (linked by resonance arrows “↔”). Include curved arrows that indicate the movement of electrons between each resonance structure. Assign non-zero formal charges to each atom for each resonance structure. (a.) NO3– (nitrate) (b.) CH3COO– (acetate) (c.) N3– (azide) (d.) NCO– (isocyanate) Problem (#3.) For each ion in question 2, draw a resonance hybrid, assigning non-zero formal and/or partial charges (δ+, δ–) as needed. Problem (#4.) For each skeletal structure below, satisfy the valences (or octets) of all of the atoms by filling in double and triple bonds as well as unshared electron pairs. Assign non-zero formal charges and show the overall charge if the structure is an ion. See photo attached for Problem number 4. Problem (#5.) For each structure in question 4, draw a resonance hybrid (if it has one) and assign non-zero formal and/or partial charges as needed.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning