
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter F, Problem 4PP
Interpretation Introduction
Interpretation:
The compound British Anti – Lewisite (BAL) is to be synthesised from allyl alcohol.
Concept introduction:
The compound British Anti – Lewisite (BAL) is used as inorganic mercury, which is effective in protecting the kidney from 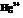 . In BAL, both the thiol groups
. In BAL, both the thiol groups 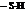 lose hydrogen and binds with the
lose hydrogen and binds with the 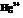 .
.
Addition of bromine across the double bond is done via electrophilic addition reaction. This can be shown as follows:
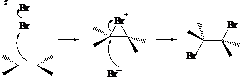
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Provide a detailed step-by-step mechanism for the reaction below. Show each intermediate species. Use arrows to show the flow of electrons and clearly indicate any atoms with formal charges.
I need help with this practice problem, thank you!
choose correct option and give detail explanation for correct and incorect answer
Which of the following are molecular formulas? Select all that apply.
Group of answer choices
C3H6O3
C2H6O
C2H4O2
C3H9O2
CH2O
Chapter F Solutions
Organic Chemistry
Knowledge Booster
Similar questions
- Which of these structures fit the following descriptions? Select all that are correct.arrow_forward10. Question shown in photo Vanillin (Figure 5, C8H8O3) is an organic compound extracted from the vanilla bean and is used to add flavor to drinks, food, and pharmaceuticals. While natural vanilla extract is composed of many different compounds, artificial vanillin is usually synthesized as a pure compound. As of 2001 only about 15% of the annual demand for vanillin was isolated as a natural product. Identify the hybridization of the labeled atoms in vanillin. a) i (sp2 ); ii (sp2 ); iii (sp2 ) b) i (sp2 ); ii (sp2 ); iii (sp3 ) c) i (sp2 ); ii (sp3 ); iii (sp2 ) d) i (sp3 ); ii (sp2 ); iii (sp2 )arrow_forwardScenario Four Read the following extract: “A polymer is a substance which has a molecular structure comprising of a large number of identical molecules covalently bonded together to form a long chain structure. Many synthetic organic materials are polymers, such as nylon, polythene and Kevlar.” The extract mentions ‘covalent bonds’. Explain what these are, and the formation of different types that can exist i.e. single, multiple etc. Do some research and identify 4 simply covalently bonded molecules. Draw dot and cross diagrams of each to show the bond formation of each of your 4 examples.arrow_forward
- Question from at-home practice: The unknown is C6H12O2. The number of rings and/or π-bonds present in the unknown can be calculated using the formula shown in the image below. Calculate the number of rings and/or p-bonds in your unknown (also known as index of hydrogen deficiency index).arrow_forwardHow many straight-chain isomers can you write that fit the following descriptions? See Worked Example 12.12 for guidance.(a) Alcohols (-OH) with a longest chain of 6 carbons(b) Amines (-NH2)with a longest chain of 7 carbonsarrow_forwardGive explanation answerarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning