9. Shown below is a binding pocket for a protein with a ligand bound. The ligand interacts with a serine and a valine side chain. HN ligand a. Briefly explain, in terms of Kd, the effect of a mutation that replaces the serine residue with a valine residue. b. Briefly explain, in terms of Kd, the effect of a mutation that alters the ligand (as seen below) binding to the original, unmutated, binding site. :0: ligand 2
9. Shown below is a binding pocket for a protein with a ligand bound. The ligand interacts with a serine and a valine side chain. HN ligand a. Briefly explain, in terms of Kd, the effect of a mutation that replaces the serine residue with a valine residue. b. Briefly explain, in terms of Kd, the effect of a mutation that alters the ligand (as seen below) binding to the original, unmutated, binding site. :0: ligand 2
Biology: The Dynamic Science (MindTap Course List)
4th Edition
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Chapter6: Energy, Enzymes, And Biological Reactions
Section: Chapter Questions
Problem 8TYK: Which of the following statements about the allosteric site is true? a. The allosteric site is a...
Related questions
Question
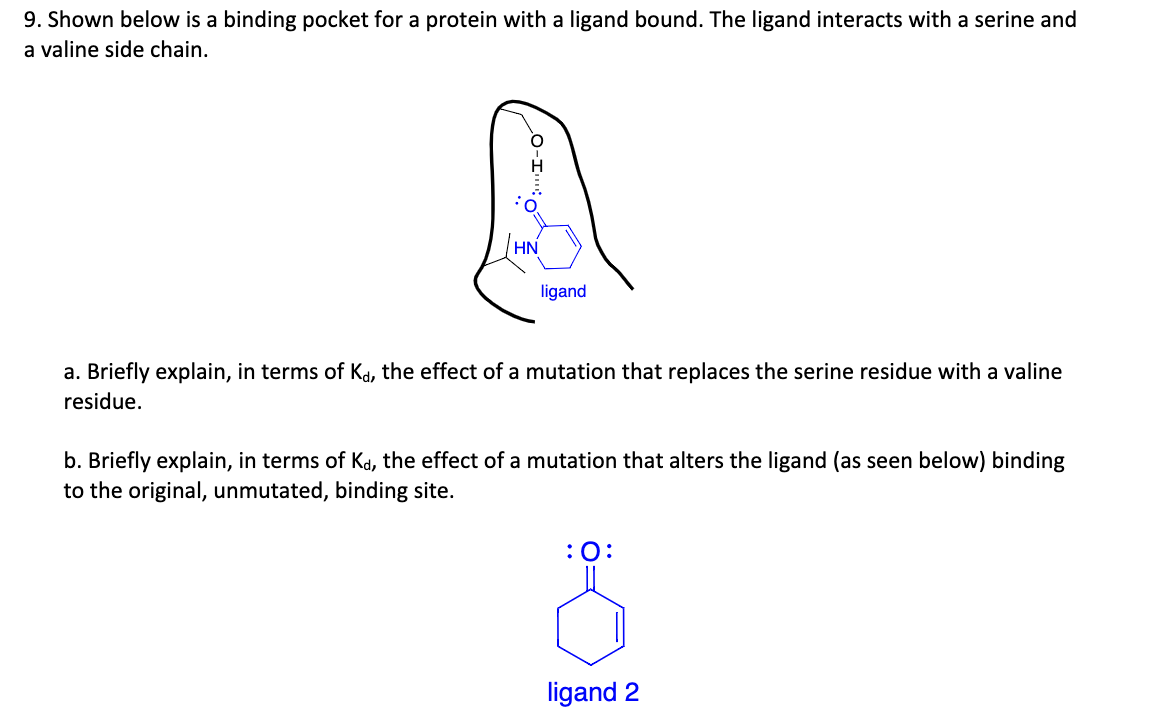
Transcribed Image Text:9. Shown below is a binding pocket for a protein with a ligand bound. The ligand interacts with a serine and
a valine side chain.
ligand
a. Briefly explain, in terms of Ka, the effect of a mutation that replaces the serine residue with a valine
residue.
b. Briefly explain, in terms of Kd, the effect of a mutation that alters the ligand (as seen below) binding
to the original, unmutated, binding site.
:0:
ligand 2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning