Prepare standard solutions for the spectrophotometric measurement of iron in an unknown solution. The first standard solution includes 250 mg/L of pure iron within 100 mL solution. Make a serial dilution by pipetting 60 mL of the first standard and the last standard solution will contain 19,44 mg/L of pure iron. wwm www wm ww m m wwww.v wwww wwww a. Complete number of standards and indicate concentration of each standard solution wwww ww wwwwww wwm w STD-X STD-1 (250 mg L) (19,44 mgL) b. If the standard curve equation is given as y=2548X-654, calculate the iron concentration of a solution that has a 0,898 absorbance value at 525 nm wavelength. wwww v
Prepare standard solutions for the spectrophotometric measurement of iron in an unknown solution. The first standard solution includes 250 mg/L of pure iron within 100 mL solution. Make a serial dilution by pipetting 60 mL of the first standard and the last standard solution will contain 19,44 mg/L of pure iron. wwm www wm ww m m wwww.v wwww wwww a. Complete number of standards and indicate concentration of each standard solution wwww ww wwwwww wwm w STD-X STD-1 (250 mg L) (19,44 mgL) b. If the standard curve equation is given as y=2548X-654, calculate the iron concentration of a solution that has a 0,898 absorbance value at 525 nm wavelength. wwww v
Chapter27: Molecular Fluorescence Spectroscopy
Section: Chapter Questions
Problem 27.13QAP
Related questions
Question
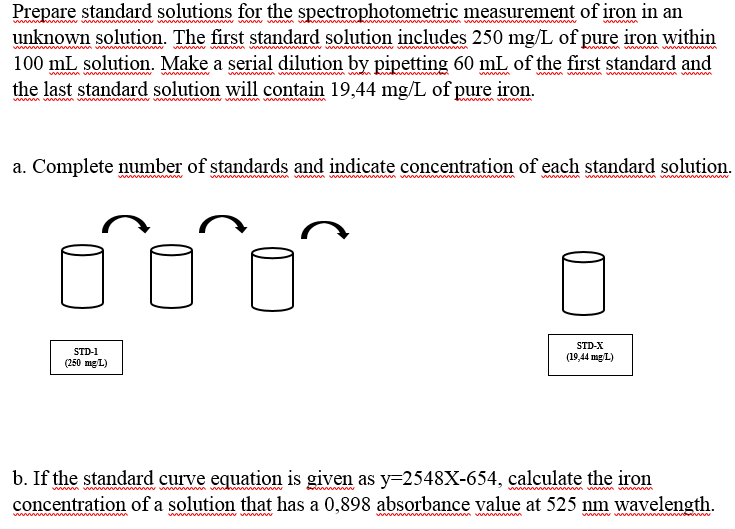
Transcribed Image Text:Prepare standard solutions for the spectrophotometric measurement of iron in an
unknown solution. The first standard solution includes 250 mg/L of pure iron within
100 mL solution. Make a serial dilution by pipetting 60 mL of the first standard and
the last standard solution will contain 19,44 mg/L of pure iron.
www w
ww ww wm
www
wwww wwwm www
a. Complete number of standards and indicate concentration of each standard solution.
www
wwwww wwwww vww vwwwwwwww vw w
w m w m ww
STD-X
STD-1
(19,44 mg L)
(250 mg L)
b. If the standard curve equation is given as y=2548X-654, calculate the iron
concentration of a solution that has a 0,898 absorbance value at 525 nm wavelength.
www
www w
www
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

