
Concept explainers
The halogen atom of an
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK ORGANIC CHEMISTRY
Additional Science Textbook Solutions
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry: A Molecular Approach (4th Edition)
Organic Chemistry (8th Edition)
Chemistry: A Molecular Approach
Chemistry: Structure and Properties
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
- A minor route for ozone destruction in the ozone hole involves Mechanism II with bromine as X' and chlorine as X (or vice-versa). The CIO and BrO free radical molecules produced in these processes then collide with each other and rearrange their atoms to eventually yield O, and atomic chlorine and bromine. Write out the mechanism for this process, and add up the steps to determine the overall reaction.arrow_forwardThe overall reaction of hydroxide radical with hydrogen gas occurs as follows: OH'g) + H,(g) – H0(g) + H'(g) Part A This reaction plays a significant role in the chemistry of the earth's atmosphere by eventually converting OH to HO, through the subsequent reaction of H with O,. HO, is involved in the destruction of stratospheric ozone (J. Phys. Chem. A, 2006, 110, 6978). Determine the activation energy for the reaction of OH with H, reaction. The rate constants for the reaction of OH with H, were found to be 5.54x10 L mol s at 36. C and 1.09x10 L molrs at -27. °C. -20.9 kJ mol1 O 18.3 kJ mol1 O 25.1 kJ mol O -18.3 kJ mol O 25.1 kJ molarrow_forwardThe compound below is treated with chlorine in the presence of light. CH3 CH,CHCH3 Draw the structure for the organic radical species produced by reaction of the compound with a chlorine atom. Assume reaction occurs at the weakest C-H bond. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms.arrow_forward
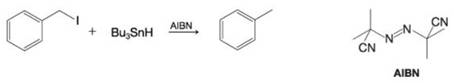
- The compound below is treated with chlorine in the presence of light. H₂C CH3 H₂C CH3 Draw the structure for the organic radical species produced by reaction of the compound with a chlorine atom. Assume reaction occurs at the weakest C-H bond. You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. Undoarrow_forwardAlkyl halides can be reduced to alkanes by a radical reaction with tributyltin hydride, (C4H9)3SnH, in the presence of light (hv). Propose a radical chain mechanism by which the reaction might occur. The initiation step is the light-induced homolytic cleavage of the Sn-H bond to yield a tributyltin radical.arrow_forwardThe compound below is treated with chlorine in the presence of light. CH3 CH3CHCH,CH3 Draw the structure for the organic radical species produced by reaction of the compound with a chlorine atom. Assume reaction occurs at the weakest C-H bond. • You do not have to consider stereochemistry. You do not have to explicitly draw H atoms.arrow_forward
- The reaction below represents what phenomenon? H,O+hv → HO•+H formation of the hydroxyl radical in the relatively unpolluted troposphere the formation of hydroxyl radical at higher altitudes destruction of the hydroxyl radical at higher altitudes the strong reactivity of the hydroxyl radicalarrow_forwardThis reaction does not take place. All that happens under experimental conditions for the formation of radicals is initiation to form iodine radicals, I·, followed by termination to reform I2. How do you account for these observations?arrow_forwardDescribe the reaction to form the catalyst L-prolinamide. (reaction is added below) .arrow_forward
- Consider the following structures: CH, CH3 H;C, „NH3 H,C, CH H3C, Br H3C. CH CH3 CH3 CH3 JECE CEEJ JEEC СЕЈЕ ЕСЕ Which is a resonance contributor? ECJE Which has a net formal charge of +1? JECE Which has only one type of hydrogen? ECJE Which will form the most stable free radical? JEEC Which is an electrophile? JEECarrow_forwardDetermine the order of the reaction with respect to H2. O 3rd order O Oth order O 2nd order O 1st orderarrow_forwardWhy does the first reaction, but not the second, give ring expansion?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

