
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
2nd Edition
ISBN: 9780077633707
Author: Janice Smith
Publisher: Mcgraw-hill Higher Education (us)
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 12, Problem 12.35UKC
Consider the following ball-and-stick model of an alcohol.
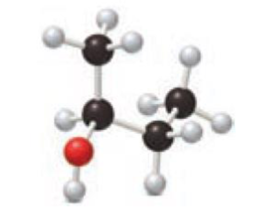
- a. Locate the chirality center.
- b. Classify the alcohol as 1°, 2°, or 3°.
- c. Name the alcohol.
- d. Draw the structure of the product formed when the alcohol is oxidized.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1.
Which compounds contain chiral centers? Show your solution for each
ww ww w w
item.
a. 2-Chloropentane
b. 3-Chloro-1-pentene
c. 3-Chloropentane
d. 1,2-Dichloropropane
1. Classify the molecule shown according to the location of its carbonyl group and the
number of carbon atoms.
CH₂OH
C=O
PIL
a. Aldotriose
b. Aldotetrose
Aldopentose
d., Ketotriose
Ketotetrose
HO-C-H
L
CH₂OH
28. A structural feature with in a molecule
which is responsible for chirality is called
c. chirality
d. Chiral C
e. Chiral center
d. all
29. Optically active compounds have isomer
a Two
C. four
b. Three
d. five
30. Propanone and propenol are
a. structural isomers
C. tautomer
b. metamers
d position isomer
Chapter 12 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
Ch. 12.1 - a. Label the hydroxyl groups, thiols, halogens,...Ch. 12.1 - Draw out each compound to clearly show what groups...Ch. 12.2 - Classify each alcohol as 1, 2, or 3.Ch. 12.2 - Classify each hydroxyl group in sorbitol as 1, 2,...Ch. 12.2 - Which compound in each pair has the higher boiling...Ch. 12.2 - Label each compound as water soluble or water...Ch. 12.2 - Give the IUPAC name for each compound.Ch. 12.2 - Give the structure corresponding to each name. a....Ch. 12.3 - Name each ether. a. CH3OCH2CH2CH2CH3 b....Ch. 12.3 - Prob. 12.10P
Ch. 12.3 - Which compound in each pair has the higher boiling...Ch. 12.5 - Prob. 12.12PCh. 12.5 - Prob. 12.13PCh. 12.6 - Prob. 12.14PCh. 12.6 - Prob. 12.15PCh. 12.6 - Give the structure corresponding to each name. a....Ch. 12.7 - Prob. 12.17PCh. 12.8 - Give the IUPAC name for each aldehyde. a....Ch. 12.8 - Prob. 12.19PCh. 12.8 - Give the IUPAC name for each aldehyde depicted in...Ch. 12.8 - Prob. 12.21PCh. 12.8 - Prob. 12.22PCh. 12.8 - Acetone and progesterone are two ketones that...Ch. 12.9 - Prob. 12.24PCh. 12.10 - Prob. 12.25PCh. 12.11 - Prob. 12.26PCh. 12.11 - Prob. 12.27PCh. 12.11 - Prob. 12.28PCh. 12.11 - Prob. 12.29PCh. 12.11 - Prob. 12.30PCh. 12.11 - Prob. 12.31PCh. 12.11 - Prob. 12.32PCh. 12 - Prob. 12.33UKCCh. 12 - Prob. 12.34UKCCh. 12 - Consider the following ball-and-stick model of an...Ch. 12 - Consider the following ball-and-stick model. a....Ch. 12 - Name each compound. a. CH3CH2OCH2CH2CH2CH3Ch. 12 - Name each compound. a. CH3OCH2CH2CH3 b....Ch. 12 - Answer the following questions about alcohol A. a....Ch. 12 - Answer the following questions about alcohol B. a....Ch. 12 - Prob. 12.41UKCCh. 12 - Prob. 12.42UKCCh. 12 - Prob. 12.43UKCCh. 12 - Prob. 12.44UKCCh. 12 - Prob. 12.45APCh. 12 - Prob. 12.46APCh. 12 - Prob. 12.47APCh. 12 - Prob. 12.48APCh. 12 - Prob. 12.49APCh. 12 - Prob. 12.50APCh. 12 - Prob. 12.51APCh. 12 - Prob. 12.52APCh. 12 - Prob. 12.53APCh. 12 - Give the structure corresponding to each name. a....Ch. 12 - Prob. 12.55APCh. 12 - Draw structures for the four constitutional...Ch. 12 - Prob. 12.57APCh. 12 - Rank the following compounds in order of...Ch. 12 - Explain why two four-carbon organic molecules have...Ch. 12 - Explain why the boiling point of CH3CH2CH2CH2OH...Ch. 12 - Which compound in each pair has the higher boiling...Ch. 12 - Which compound in each pair is more water soluble?...Ch. 12 - Prob. 12.63APCh. 12 - Prob. 12.64APCh. 12 - Prob. 12.65APCh. 12 - Prob. 12.66APCh. 12 - Prob. 12.67APCh. 12 - Xylitol is a nontoxic compound as sweet as table...Ch. 12 - Prob. 12.69APCh. 12 - Prob. 12.70APCh. 12 - Prob. 12.71APCh. 12 - Prob. 12.72APCh. 12 - Prob. 12.73APCh. 12 - Prob. 12.74APCh. 12 - Prob. 12.75APCh. 12 - Prob. 12.76APCh. 12 - Prob. 12.77APCh. 12 - Draw the structure corresponding to each name. a....Ch. 12 - Prob. 12.79APCh. 12 - Prob. 12.80APCh. 12 - What product is formed when each compound is...Ch. 12 - Prob. 12.82APCh. 12 - Prob. 12.83APCh. 12 - Prob. 12.84APCh. 12 - Prob. 12.85APCh. 12 - Prob. 12.86APCh. 12 - Prob. 12.87APCh. 12 - Label each of the following objects as chiral or...Ch. 12 - Prob. 12.89APCh. 12 - Prob. 12.90APCh. 12 - Prob. 12.91APCh. 12 - Prob. 12.92APCh. 12 - Prob. 12.93APCh. 12 - Prob. 12.94APCh. 12 - Prob. 12.95APCh. 12 - Prob. 12.96APCh. 12 - Prob. 12.97APCh. 12 - How are the compounds in each pair related? Are...Ch. 12 - Prob. 12.99APCh. 12 - Prob. 12.100APCh. 12 - Prob. 12.101APCh. 12 - Prob. 12.102APCh. 12 - Prob. 12.103APCh. 12 - Lactic acid [CH3CH(OH)CO2H] gives sour milk its...Ch. 12 - Prob. 12.105APCh. 12 - Prob. 12.106APCh. 12 - Prob. 12.107CPCh. 12 - Prob. 12.108CPCh. 12 - Prob. 12.109BTCCh. 12 - Prob. 12.110BTC
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. Assign a name to each of the following monosaccharides. Use D and L designations. H I CH 2OH C=O C=O a. b. a. HO-C-H H-C-OH H-C-OH CH₂OH d. True or False: b. HỌCH CH ₂OH C. Circle all chiral centers for the monosaccharides shown. The molecules shown can rotate plane polarized light?arrow_forwardIdentify the organic functional groups and reaction type for the following reaction.The reactant is a(n)a. two hemiacetalsb. disaccharidec. esterd. ethere. alcohol pyranosesThe product is a(n)a. beta 1-6 disaccharideb. beta 1-4 disaccharidec. alpha 1-6 disaccharided. beta 1-2 disaccharidee. alpha 1-2 disaccharidef. alcohol pyranosesg. alpha 1-4 disaccharideh. two hemiacetalsThe reaction type is:a. hydrolysisb. mutarotationc. reduction (hydrogenation)d. hemiacetal formatione. acetal formationf. oxidation (benedict's)arrow_forwardstructure of each type of compound. A. D –aldotriose B. L-ketohexosearrow_forward
- 12. Monosaccharides are designated with a "D" or "L" in their name. What determines this? A. The bond between a chiral carbon and an OH group in the structure. B. The acyclic monosaccharide contains either a ketone or an aldehyde functional group. C. The molecule has either five or six carbons in its structure. D. The molecule is either cyclic or acyclic. E. The anomeric carbon has an OH group pointing up or down in a drawing of the molecule.arrow_forward7. Considering the following chemical, which statement is true? CHO H-C-OH HO-C-H HO-C-H H-C-OH CH₂OH A. It needs to form the ring form first before it can interact with methanol to form an intramolecular hemiketal B. When the molecule forms the ring form, the carbon atom #5 becomes the anomeric carbon C. Both A and B D. Neither A nor Barrow_forwardClassify the following disaccharide. a. beta 1-2 disaccharide b. alpha 1-4 disaccharide c. alpha 1-6 disaccharide d. alpha 1-2 disaccharide e. beta 1-4 disaccharide f. beta 1-6 disaccharidearrow_forward
- Which of the following types of carbohydrates can be hydrolyzed to give smaller molecules? I. monosaccharides II. disaccharides II. oligosaccharides IV. polysaccharides 7. a.l only b.Il and IV only c. II only d.l and II only e. II, III, and IVarrow_forwardWhich of the following is not a physical property of alcohols or phenols? A. The solubilities of primary alcohols in water decrease with increasing molecular weight. B. Due to hydrogen bonding, boiling points of alcohols is much higher than those of corresponding alkanes. C. The hydroxyl group of an alcohol is nonpolar. D. Phenols are generally only slightly soluble in water.arrow_forward1. When an aldehyde is reduced with hydrogen (H2) gas in the presence of a transition metal catalyst, what type of product is formed? A. primary alcohol B. secondary alcohol C. tertiary alcohol D. carboxylic acidarrow_forward
- 33. Determine whether or not each molecule is a fatty acid. If it is a fatty acid, classify it as saturated, monounsaturated, or polyun- saturated. a. CH HO, b. CH, CH CH, . CH CH;-C-OH d. CH, CH=CHarrow_forward1. Use asterisks to show the chiral center(s) in the following structures: CHO a. HO b. H CH2OH H OH H 0 H OH H OH b. H H. + OH • OH OH CH₂OH 2. Name the monosaccharides show in question 1. Be as specific as possible a.arrow_forwardWhat functional groups are present in a carbohydrate molecule? a. Carboxyl and carbonyl groups b. Alcohol and carboxyl groups c. Hydroxyl and carbonyl groups d. Hydroxyl and hydrogen groupsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY