
Concept explainers
a.
Interpretation:
The product obtained when the below compound reacts with H2SO4 reagent has to be determined.
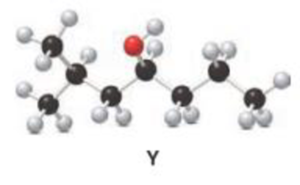
Concept introduction:
Dehydration of alcohol:
When alcohol reacts with
b.
Interpretation:
For a given Y is treated with
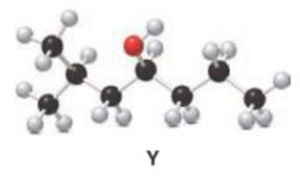
Concept introduction:
Oxidation of alcohol:
During oxidation of alcohol, the number of
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- Draw the products formed when cholesterol is treated with each reagent. Indicate the stereochemistry around anystereogenic centers in the product.a. CH3COCIb. H2, Pd-Cc. PCCd. leic acid, H+e. [1] BH3 ·THF; [2] H2O2, -OHarrow_forwardDraw the products formed when CH3CH2C ≡ CCH2CH3 is treated with each reagent: (a) Br2(2 equiv); (b) Cl2 (1 equiv).arrow_forwardH CH3 CH3 H3C- H3C- CH3 CH3 (1) a. Identical Enantiomers Diastereoisomers Structural isomers piinoo etuload CH3 Et H Et H CH3 Et H3C H. b. ČH3 Et 翠 Identical Enantiomers Diastereoisomers Structural isomersarrow_forward
- Draw the organic product(s) formed when CH3CH2CH2OH is treated withfollowing reagent : [1] TsCl, pyridine; [2] NaSHarrow_forward(d) Draw the structure of the expected product when monosaccharide B undergo mutarotation upon dissolving in water in the presence of Tollens reagent (AGNO3, NHẠOH). он OH O. OH OH OH monosaccharide Barrow_forwardH₂O H2SO4 (cat.)arrow_forward
- a. Reaction of acyl compounds with LiAlH4 forms alcohols. b.Reaction of acyl compounds with alcohols forms esters. c.Reaction of carbonyl compounds with LiAlH4 forms alcohols. d.Reaction of carbonyl compounds with alcohols forms hemiacetals. Which are false?arrow_forwardDraw the structure of each type of compound. a. a D-aldotriose b. an L-ketohexose c. a four-carbon aldonic acidarrow_forward— 9 Give the organic product: C. CH₂CH₂CH₂-C CH -NH₂ 1) NINH, 2) CH₂CH₂I B. D.arrow_forward
- Draw the organic product(s) formed when CH3CH2CH2OH is treated withfollowing reagent : TsCl, pyridinearrow_forwardDraw the products formed when ß-D-galactose is treated with each reagent.a. Ag2O + CH3Ib. NaH + C6H5CH2Clc. The product in (b), then H3O+d. Ac2O + pyridinee. C6H5COCl + pyridinef. The product in (c), then C6H5COCl + pyridinearrow_forwardConsider the tetrasaccharide stachyose drawn below. Stachyose is found in white jasmine,soybeans, and lentils. Because humans cannot digest it, its consumption causes flatulence. a. Is stachyose a reducing sugar?b. What product is formed when stachyose is treated with excess CH3I, Ag2O?c. What products are formed when the product in (e) is treated with H3O+?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





