
Interpretation:
For the below compound the chirality centers has to be labeled and number of chiral centers and the type of alcohol should be determined.
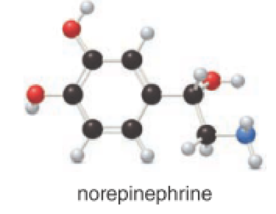
Concept introduction:
Chirality center:
When a carbon atom is bonded to four different groups, then that carbon is named as chiral carbon and it is called as chirality center. Generally, the carbon atom contains tetrahedral valency, therefore it bonded to four atoms or molecules. The chiral center is bonded with four different elements or groups. If the single carbon atom is bonded to two or more identical group or atom, then it is also not considered as chiral carbon. In addition to this, if the carbon atom contains multiple bond then it does not able to bond with four different groups then it is not considered to be as a chirality center.
Classification of alcohol:
Generally the alcohol group is bonded with minimum one alkyl group. The alcohol group is classified into three types, such as primary
Explanation of Solution
Given compound is norepinephrine,
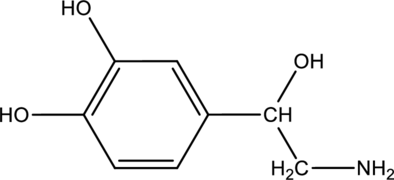
In the given drug (i,e) norepinephrine, the
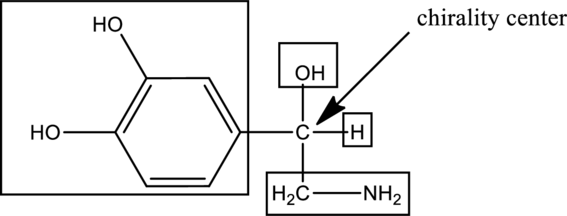
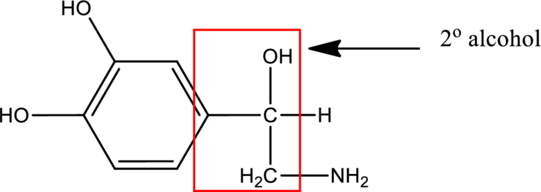
In the below compound,
Want to see more full solutions like this?
Chapter 12 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- List the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forward18-19 The following compounds have approximately the same molecular weight: hexanoic acid, heptanal, and 1-heptanol. Arrange them in order of increasing boiling point.arrow_forwardWhy is it safe for us to consume foods like vinegar that contain acetic acids?arrow_forward
- 16-19 2-Me thy 1 propane (bp -12°C), 2-propanol (bp 82°C), and 2-propanamine (bp 32°C) all have approximately the same molecular weight, yet their boiling points are quite different. Explain the reason for these differences.arrow_forwardDraw structural formulas for the following entities. a. Propanoate ion b. Sodium propanoate c. Acetate ion d. Sodium acetatearrow_forward14-55 (Chemical Connections 140 The legal definition of being under the influence of alcohol is based on blood alcohol content. What is the relationship between breath alcohol content and blood alcohol content?arrow_forward
- 16-54 Several poisonous plants, including Atropa belladonna, contain the alkaloid atropine. The name “belladonna” (which means “beautiful lady”) probably comes from the fact that Roman women used extracts from this plant to make themselves more attractive. Atropine is widely used by ophthal mologists and optometrists to dilate the pupils for eye examination. Classify the amino group in atropine as primary, secondary, or tertiary. Locate all stereocenters in atropine. Account for the fact that atropine is almost insoluble in water (1 g in 455 mL of cold water) but atropine hydrogen sulfate is very soluble (1 g in 5 mL of cold water). Account for the fact that a dilute aqueous solution of atropine is basic (pH approximately 10.0).arrow_forwardLabel each of the following structures as a hemiacetal, hemiketal, acetal, ketal, or none of these: a. c. b.arrow_forwardHow many kinds of chemically non-equivalent carbons are there in each of the following compounds? a b علم CH₂-CH₂- +NH₂ OH CH₂ Aspirin (analgesic, antipyretic and antibiotic) The number of chemically non-equivalent carbons is The number of chemically non-equivalent carbons isarrow_forward
- Draw the eight constitutional isomers with molecular formula C5H120 that contain an OH group. Give the IUPAC name for each compound. Classify each alcohol as 1°, 2°, or 3°.arrow_forwardTHC is the active component in marijuana, and ethanol is the alcohol in alcoholic beverages. Explain why drug screenings are able to detect the presence of THC but not ethanol weeks after these substances have been introduced into the body.arrow_forwardThe following molecule is a NH 3° amine 2° alcohol 1° alcohol 2° amine 1° amine 3° alcoholarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning



