
a.
Interpretation:
For a given alcohol compound is
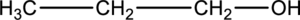
Concept Introduction:
Classification of alcohol:
Generally, the alcohol group is bonded with minimum one alkyl group. The alcohol group is classified into three types, such as primary
b.
Interpretation:
For a given alcohol compound is
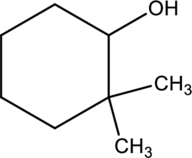
Concept introduction:
Refer to part a.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- 18. Ketone reduction Dicyclohexyl ketone Reduce the ketone. 1. NaBH4, ethanol 2. H3O+ H OH Dicyclohexylmethanol (88%) (a 2° alcohol)arrow_forwardDiethyl ether and butan-1-ol are isomers, and they have similar solubilities in water. Their boiling points are very different, however. Explain why these two compounds have similar solubility properties but dramatically different boiling points. CH3CH2¬O¬CH2CH3 CH3CH2CH2CH2¬OH diethyl ether, bp 35 °C butan-1-ol, bp 118 °C 8.4 mL dissolves in 100 mL H2O 9.1 mL dissolves in 100 mL H2Oarrow_forwardClassify the carbon atoms in each compound as 1°, 2°, 3°, or 4°. a. CH3CH2CH2CH3 b. (CH3)3CHarrow_forward
- What products are formed when each alcohol is oxidized with K2Cr2O7? a. CH3CH2CH2CH2CH2OHarrow_forwardWhich alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3larrow_forward3. Complete the following intramolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) Cy-C-CH-oH 18UC b) CHy CH-Cy-CHy 180C OH c) 180c 4. Complete the following intermolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) CHy-CH-OH b) Ho SわふわSわ 5. Complete the following oxidation reactions for alcohols. Draw the structure of the product. Name the reactant and identify the type of compound formed in the product. a) hparrow_forward
- 3) Identify the compound with the lowest boiling point. A. CH3CH2CH2OH B. CH:CH(OH)CH3 C. CH3OCH2CH3 D. CH3CH2CH2CH2OH E. CH3CH2OCH2CH3arrow_forwardWhich of these compounds forms hydrogen bonds with a solvent such as ethanol? 1. CH3CH2CH2OH 2. CH3CH2CH2F 3. CH2OCH2CH3arrow_forward1. Why 70% alcohol is used as disinfectant? 2. Why pure concentrated alcohol is not 100%? 3. What are the symptoms of alcohol intoxication?arrow_forward
- Organic Reaction Write an equation for the oxidation of each alcohol. Use [O] above the arrow to indicate an oxidizing agent. If no reaction occurs, write "no reaction" after the arrow. 1. CH3CH2CH2CH2CH2OH CH₂ CH₂CCH₂CH₂ ОН CH CHCH CH CH CH, ОНarrow_forwardClassify these alcohols as primary (1°), secondary (2°), or tertiary (3°). 1° 2° 3° CH,CH, CH,COH ČH,CH, OH CH,CHCHCH, CH, CH,CH, CH,CCH,OHarrow_forwardH3C. H3C CH3 Br 1-propanolarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
