
Concept explainers
a.
Interpretation:
Product formed when below alcohol is oxidized in presence of K2Cr2O7 has to be determined.
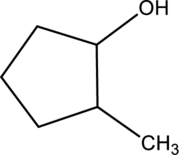
Oxidation of alcohol:
During oxidation of alcohol, the number of
b.
Interpretation:
Product formed when below alcohol is oxidized in presence of K2Cr2O7 has to be determined.
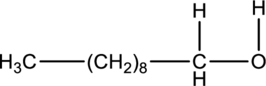
Concept introduction:
Refer to part ‘a.’.
c.
Interpretation:
Product formed when below alcohol is oxidized in presence of K2Cr2O7 has to be determined.
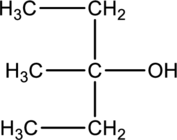
Concept introduction:
Refer to part ‘a.’.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- What alcohol is formed when the compound is treated with H2 and a Pd catalyst?arrow_forward3. Complete the following intramolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) Cy-C-CH-oH 18UC b) CHy CH-Cy-CHy 180C OH c) 180c 4. Complete the following intermolecular dehydration reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) CHy-CH-OH b) Ho SわふわSわ 5. Complete the following oxidation reactions for alcohols. Draw the structure of the product. Name the reactant and identify the type of compound formed in the product. a) hparrow_forward8. Give the IUPAC name for each alkene. CH3 CH2CH,CH3 Br а.arrow_forward
- Label each ether and alcohol in brevenal, a marine natural product. Classify each alcohol as 1°, 2°, or 3°.arrow_forward4) A reagent that can reduce a carboxylic acid to an alcohol is A) H2/Pt B) K2Cr207 C) NaBH4 D) LiAlH4arrow_forwardGive the IMPAC name for each compound. 1. COOH COOH 2. Br 3. CH3 CHz COOHarrow_forward
- What product is formed when the alcohol is oxidized with K2Cr2O7? In some cases, no reaction occurs (if so, draw the given alcohol).arrow_forwardLabel each ether and alcohol in brevenal, a marine natural product. Classify each alcohol as 1°, 2°, or 3°.arrow_forwardDraw the structures of the chief product formed when the following alcohols are dehydrated to alkenes: ОН CH, a. CH, ОН b. CH,ССH,CHСH, CH3arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning  Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





