
Concept explainers
(a)
Interpretation:
The IUPAC name of the given compound needs to be determined.
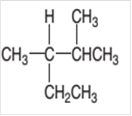
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with
(b)
Interpretation:
The IUPAC name of the given compound needs to be determined.
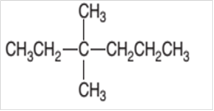
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with chemical bonds and the arrangement of atoms in the molecule. IUPAC purposed some rules to determine the name of organic compound that is based on the number of C atoms in the longest chain of the compound and name of branches.
(c)
Interpretation:
The IUPAC name of the given compound needs to be determined.
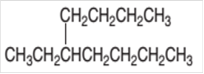
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with chemical bonds and the arrangement of atoms in the molecule. IUPAC purposed some rules to determine the name of organic compound that is based on the number of C atoms in the longest chain of the compound and name of branches.
(d)
Interpretation:
The IUPAC name of the given compound needs to be determined.
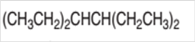
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with chemical bonds and the arrangement of atoms in the molecule. IUPAC purposed some rules to determine the name of organic compound that is based on the number of C atoms in the longest chain of the compound and name of branches.
(e)
Interpretation:
The IUPAC name of the given compound needs to be determined.
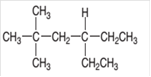
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with chemical bonds and the arrangement of atoms in the molecule. IUPAC purposed some rules to determine the name of organic compound that is based on the number of C atoms in the longest chain of the compound and name of branches.
(f)
Interpretation:
The IUPAC name of the given compound needs to be determined.
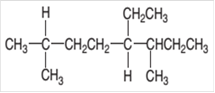
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols. The structural formula represents all the bonded atoms with chemical bonds and the arrangement of atoms in the molecule. IUPAC purposed some rules to determine the name of organic compound that is based on the number of C atoms in the longest chain of the compound and name of branches.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Why can formaldehyde (CH20) be prepared in the form of a 37% solution in water, whereas octanal cannot? (Select all that apply.) | Octanal is more hydrophilic than formaldehyde. O Formaldehyde is a small molecule. O Formaldehyde is polar. O Octanal is mostly hydrophobic.arrow_forwardThe atomic radii of elements when one moves from right to left in a period Increase Decrease Remain stable Fluctuate According to the quinonoid theory, in the benzenoid form indicators show- Light colour None of the above No color O Deep colorarrow_forwardDraw the eight constitutional isomers with molecular formula C5H120 that contain an OH group. Give the IUPAC name for each compound. Classify each alcohol as 1°, 2°, or 3°.arrow_forward
- Give the IUPAC name for the following compound. OH 4-ethyl-2-methyl-3-heptanol 3-ethyl-2-methyl-4-heptanol None of the choices are correct ○ 4-ethyl-2-methyl-3-octanol 4-ethyl-6-methyl-5-heptanolarrow_forwardWhat is the IUPAC name of the ether that has the structure shown? -O-CH3arrow_forwardDraw structures for the four constitutional isomers of molecular formula C4H10O that contain an OH group. Give the IUPAC name for each alcohol.arrow_forward
- What is the IUPAC name of the following molecule? OH O 3-ethyl-4-sec-hexyl-hexan-3-ol O 3,4,5-triethyloctan-3-ol 3,4,5-triethyloctanol O 3,4-diethyl-4-propylheptan-3-olarrow_forwardWhat alcohol is formed when each compound is treated with NaBH4 in CH3OH?arrow_forwardThe correct IUPAC name for CH3-CH(OH) CH2 CH2-CH2-CH2(OH) is a) hexyl dialcohol b) 2,6-dihydroxyhexane c) 1,5-hexanediol d) 1,6-hexanediolarrow_forward
- Which of the following alcohols is the MOST soluble in water? O 2-methyl-2-pentanol O 4-methyl-2-pentanol O 3-methyl-1-butanol O 2-methyl cyclohexanolarrow_forwardDraw four constitutional isomers having molecular formula C 6H 12 that contain a four-membered ring. Give the IUPAC name for each isomer.arrow_forwardGive the IUPAC name for each structure. Part 1 of 3 CH3(CH2)2CO₂ (CH2)CH3 Part 2 of 3 Part 3 of 3 -CO,—CH,—CH,arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

