
Concept explainers
(a)
Interpretation:
The starting material required to prepare the given
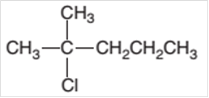
Concept Introduction:
In a
The conversion of reactant to product is represented by an arrow placed between them.
Halogenation reaction is a substitution reaction in which one of the H atom of reactant hydrocarbon is substituted with halogen atom to form respective alkyl halide.
(b)
Interpretation:
The starting material to prepare the given alkyl halide by a halogenation reaction needs to be determined.
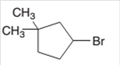
Concept Introduction:
In a chemical reaction, the conversion of a substance to new substances can be represented. The substance initially present in the reaction is reactant and the new substance formed is known as product.
The conversion of reactant to product is represented by an arrow placed between them.
Halogenation reaction is a substitution reaction in which one of the H atom of reactant hydrocarbon is substituted with halogen atom to form respective alkyl halide.
(c)
Interpretation:
The starting material to prepare the given alkyl halide by a halogenation reaction needs to be determined.
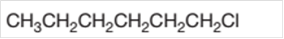
Concept Introduction:
In a chemical reaction, the conversion of a substance to new substances can be represented. The substance initially present in the reaction is reactant and the new substance formed is known as product.
The conversion of reactant to product is represented by an arrow placed between them.
Halogenation reaction is a substitution reaction in which one of the H atom of reactant hydrocarbon is substituted with halogen atom to form respective alkyl halide.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Supply the structural formula of the product in each of the following alkene addition reactions. a. CH3CH=CHCH3 + Cl2 ? b. CH3CHCH=CH2 + HCl ?arrow_forwardWhat reactant would you use to prepare each of the following compounds from cyclohexene?arrow_forwardAssign an IUPAC name to each of the following halogenated hydrocarbons. a. CH3I b. CH3CH2CH2Cl c. d.arrow_forward
- What is the molecular formula for each of the following hydrocarbons? a. 5-carbon alkene with one double bond b. 5-carbon alkene with two double bonds c. 5-carbon cycloalkene with one double bond d. 5-carbon cycloalkene with two double bondsarrow_forwardAnswer true or false. Combustion of alkanes is an endothermic reaction. The products of complete combustion of an alkane are carbon dioxide and water. Halogenation of an alkane converts it to a haloalkane.arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning



