
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
3rd Edition
ISBN: 9781259298424
Author: SMITH
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 12.85P
Answer the following questions about the 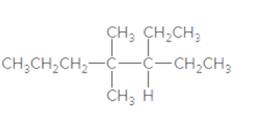
- Give the IUPAC name.
- Draw one constitutional isomer.
- Predict the solubility in water.
- Predict the solubility in an organic solvent.
- Write a balanced equation for complete combustion.
- Draw a skeletal structure.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The following names are incorrect. Draw the structure of the compound and provide the correct IUPAC name.
Molecule #1: triisobutylmethane
Molecule #2: trimethylmethane
Draw a skeleton structure and give the IUPAC name of a molecule with one alcohol group, 6 total carbons, and one methyl substituent group.
Chapter 12 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
Ch. 12.1 - How many hydrogen atoms are present in each...Ch. 12.1 - Which formulas represent acyclic alkanes and which...Ch. 12.2 - Prob. 12.3PCh. 12.2 - Draw two isomers with molecular formula C6H14 that...Ch. 12.2 - Prob. 12.5PCh. 12.2 - Classify the carbon atoms in each compound as 1°,...Ch. 12.2 - Prob. 12.7PCh. 12.2 - Prob. 12.8PCh. 12.2 - Prob. 12.9PCh. 12.2 - Prob. 12.10P
Ch. 12.4 - Give the IUPAC name for each compound.Ch. 12.4 - Give the IUPAC name for each compound....Ch. 12.4 - Prob. 12.13PCh. 12.4 - Prob. 12.14PCh. 12.5 - Prob. 12.15PCh. 12.5 - Prob. 12.16PCh. 12.5 - Give the IUPAC name for each compound.Ch. 12.5 - Prob. 12.18PCh. 12.7 - Answer the following questions about pentane...Ch. 12.7 - Prob. 12.20PCh. 12.7 - Prob. 12.21PCh. 12.8 - Prob. 12.22PCh. 12.8 - Prob. 12.23PCh. 12.9 - Prob. 12.24PCh. 12.9 - Prob. 12.25PCh. 12.9 - Prob. 12.26PCh. 12 - Prob. 12.27PCh. 12 - Prob. 12.28PCh. 12 - Prob. 12.29PCh. 12 - Prob. 12.30PCh. 12 - Classify each carbon as 1°, 2°, 3°, or 4°. a....Ch. 12 - Prob. 12.32PCh. 12 - Label each pair of compounds as constitutional...Ch. 12 - Label each pair of compounds as constitutional...Ch. 12 - Consider compounds A, B, and C. Label each pair of...Ch. 12 - Consider compounds D,E, and F. Label each pair of...Ch. 12 - Prob. 12.37PCh. 12 - Prob. 12.38PCh. 12 - Draw structures that fit the following...Ch. 12 - Draw the five constitutional isomers having...Ch. 12 - Prob. 12.41PCh. 12 - Prob. 12.42PCh. 12 - Prob. 12.43PCh. 12 - Prob. 12.44PCh. 12 - Prob. 12.45PCh. 12 - Prob. 12.46PCh. 12 - Prob. 12.47PCh. 12 - Prob. 12.48PCh. 12 - Prob. 12.49PCh. 12 - Prob. 12.50PCh. 12 - Give the IUPAC name for each cycloalkane.Ch. 12 - Prob. 12.52PCh. 12 - Prob. 12.53PCh. 12 - Give the structure corresponding to each IUPAC...Ch. 12 - Each of the following IUPAC names is incorrect....Ch. 12 - Each of the following IUPAC names is incorrect....Ch. 12 - Draw three constitutional isomers having molecular...Ch. 12 - Draw four constitutional isomers having molecular...Ch. 12 - Draw a skeletal structure for each compound octane...Ch. 12 - Convert each compound to a skeletal structure CH3(...Ch. 12 - Convert each skeletal structure to a complete...Ch. 12 - Convert each skeletal structure to a complete...Ch. 12 - Which compound in each pair has the higher melting...Ch. 12 - Which compound in each pair has the higher boiling...Ch. 12 - Branching in an alkane chain decreases surface...Ch. 12 - Explain why the boiling points of heptane [CH3( CH...Ch. 12 - Explain why hexane is more soluble in...Ch. 12 - Mineral oil and Vaseline are both mixtures of...Ch. 12 - Write a balanced equation for the combustion of...Ch. 12 - Write a balanced equation for the combustion of...Ch. 12 - Write a balanced equation for the incomplete...Ch. 12 - Prob. 12.72PCh. 12 - Prob. 12.73PCh. 12 - Prob. 12.74PCh. 12 - Prob. 12.75PCh. 12 - Prob. 12.76PCh. 12 - Prob. 12.77PCh. 12 - Prob. 12.78PCh. 12 - Prob. 12.79PCh. 12 - Prob. 12.80PCh. 12 - Prob. 12.81PCh. 12 - Prob. 12.82PCh. 12 - Prob. 12.83PCh. 12 - A major component of animal fat is tristearin, (a)...Ch. 12 - Answer the following questions about the alkane...Ch. 12 - Prob. 12.86PCh. 12 - Prob. 12.87PCh. 12 - Answer the questions in Problem 12.85 for the...Ch. 12 - Prob. 12.89CPCh. 12 - Draw the structure of the 12 constitutional...Ch. 12 - Cyclopentane has a higher boiling point than...Ch. 12 - Prob. 12.92CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the difference in bonding and in the general molecular formula between an alkene and an alkane with the same number of carbon atoms?arrow_forwardWrite the molecular formula of each alkane.arrow_forwardAccording to Table 2.10, five constitutional isomers are possible for alkanes with six carbon atoms. Write structural and condensed formulas for these isomers.arrow_forward
- How does the structure of a cycloalkane differ from that of a straight-chain or branched-chain alkane?arrow_forwardDistinguish between isomerism and resonance. Distinguish between structural and geometric isomerism. When writing the various structural isomers, the most difficult task is identifying which are different isomers and which are identical to a previously written structurethat is, which are compounds that differ only by the rotation of a carbon single bond. How do you distinguish between structural isomers and those that are identical? Alkenes and cycloalkanes are structural isomers of each other. Give an example of each using C4H8. Another common feature of alkenes and cycloalkanes is that both have restricted rotation about one or more bonds in the compound, so both can exhibit cis- trans isomerism. What is required for an alkene or cycloalkane to exhibit cis-trans isomerism? Explain the difference between cis and trans isomers. Alcohols and ethers are structural isomers of each other, as are aldehydes and ketones. Give an example of each to illustrate. Which functional group in Table 21-4 can be structural isomers of carboxylic acids? What is optical isomerism? What do you look for to determine whether an organic compound exhibits optical isomerism? 1-Bromo-1-chloroethane is optically active whereas 1-bromo-2-chloroethane is not optically active. Explain.arrow_forwardIs the general formula of a cycloalkanes the same as the general formula of an alkane, CnH2n+2? Draw any structural diagram to illustrate your answer.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning


Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License